 The
Science Notebook
The
Science NotebookHenley's Book of Formulas, Recipes and Processes
Home Terms of Use Safety Contact Us Experiment Pages Downloads Supplies Useful Links!
Henley's Twentieth Century Book of Formulas, Recipes and Processes - Pages 51-75
[51]
ALLOYS
harder metals, and less grease or oil is consequently required when they are used.
I. An anti-friction metal of excellent quality and one that has been used with success is made as follows: 17 parts zinc; 1 part copper; l 1/2 parts antimony; prepared in the following way: Melt the copper in a small crucible, then add the antimony, and lastly the zinc, care being taken not to burn the zinc. Burning can be prevented by allowing the copper and antimony to cool slightly before adding the zinc. This metal is preferably cast into the shape desired and is not used as a lining metal because it requires too great a heat to pour. It machines nicely and takes a fine polish on bearing surfaces. It has the appearance of aluminum when finished. Use a lubricating oil made from any good grade of machine oil to which 3 parts of kerosene have been added.
II. Copper, 6 parts; tin, 12 parts; lead, 150 parts; antimony, 30 parts; wrought iron, 1 part; cast iron, 1 part. For certain purposes the composition is modified as follows: Copper, 16 parts; tin, 40 parts; lead, 120 parts; antimony, 24 parts; wrought iron, 1 part; cast iron, 1 part. In both cases the wrought iron is cut up in small pieces, and in this state it will melt readily in fused copper and cast iron. After the mixture has been well stirred, the tin, lead, and antimony are added; these are previously melted in separate crucibles, and when mingled the whole mass is again stirred thoroughly. The product may then be run into ingots, to be employed when needed. When run into the molds the surface should be well skimmed, for in this state it oxidizes
rapidly. The proportions may be varied without materially affecting the results.
III. From tin, 16 to 20 parts; antimony, 2 parts; lead, 1 part; fused together, and then blended with copper, 80 parts. Used where there is much friction or high velocity.
IV. Zinc, 6 parts; tin, 1 part; copper, 20 parts. Used when the metal is exposed to violent shocks.
V. Lead, 1 part; tin, 2 parts; zinc, 4 parts; copper, 68 parts. Used when the metal is exposed to heat.
VI. Tin, 48 to 50 parts; antimony, 5 parts; copper, 1 part.
VII. (Fenton's.) Tin, with some zinc, and a little copper.
VIII. (Ordinary.) Tin, or hard pewter, with or without a small portion of antimony or copper. Without the last it is apt to spread out under the weight of heavy machinery. Used for the bearings of locomotives, etc.
The following two compositions are for motor and dynamo shafts: 100 pounds tin; 10 pounds copper; 10 pounds antimony.
83 1/2 pounds tin; 8 1/4 pounds antimony; 8 1/4 pounds copper.
IX. Lead, 75 parts; antimony, 23 parts; tin, 2 parts.
X. Magnolia Metal. This is composad of 40 parts of lead, 7 1/2 parts of antimony, 2 1/2 of tin, 1/8 of bismuth, 1/8 of aluminum, and 1/4 of graphite. It is used as an anti-friction metal, and takes its name from its manufacturer's mark, a magnolia flower.
ARGENTAN:
See German Silver, under this title.
BELL METAL.
The composition of bell metal varies considerably, as may be seen below:
I. (Standard.) Copper, 78 parts; tin, 22 parts; fused together and cast. The most sonorous of all the alloys of copper and tin. It is easily fusible, and has a fine compact grain, and a vitreous
conchoidal and yellowish-red fracture. According to Klaproth, the finest-toned Indian gongs have this composition.
II. (Founder's Standard.) Copper, 77 parts; tin, 21 parts; antimony, 2 parts. Slightly paler and inferior to No. I.
III. Copper, 80 parts; tin, 20 parts. Very deep-toned and sonorous. Used in China and India for the larger gongs, tam-tams, etc.
IV. Copper, 78 to 80 parts; tin, 22 to 20 parts. Usual composition of Chinese cymbals, tam-tams, etc.
V. Copper, 75 (= 3) parts; tin, 25
(=1) part. Somewhat brittle. In fracture, semivitreous and bluish-red. Used for church and other large bells.
VI. Copper, 80 parts; tin, 10 1/4 parts; zinc, 5 1/2 parts; lead, 4 1/4 parts. English bell metal, according to Thomson. Inferior to the last; the lead being apt to form isolated drops, to the injury of the uniformity of the alloy.
VIL Copper, 68 parts; tin, 32 parts. Brittle; fracture conchoidal and ashgray. Best proportions for house bells, and bells, etc.; for which, however, 2 of copper and 1 of tin is commonly substituted by the founders.
VIII. Copper, 72 parts; tin, 26 1/2 parts; iron, 1 1/2 parts. Used by the Paris houses for the bells of small clocks.
IX. Copper, 72 parts; tin, 26 parts; zinc, 2 parts. Used, like the last, for very small bells.
X. Copper, 70 parts; tin, 26 parts;
[52]
ALLOYS
zinc, 2 parts. Used for the bells of repeating watches.
XI. Melt together copper, 100 parts; tin, 25 parts. After being cast into the required object, it should be made redhot, and then plunged immediately into cold water in order to impart to it the requisite degree of sonorousness. For cymbals and gongs.
XII. Melt together copper, 80 parts; tin, 20 parts. When cold it has to be hammered out with frequent annealing.
XIII. Copper, 78 parts; tin, 22 parts; This is superior to the former, and it can be rolled out. For tam-tams and gongs.
XIV. Melt together copper, 72 parts; tin, 26 to 56 parts; iron 1/44 part. Used in making the bells of ornamental French clocks.
Castings in bell metal are all more or less brittle; and, when recent, have a color varying from a dark ash-gray to grayish-white, which is darkest in the more cuprous varieties, in which it turns somewhat on the yellowish-red or bluish-red. The larger the proportion of copper in the alloy, the deeper and graver the tone of the bells formed of it. The addition of tin, iron, or zinc, causes them to give out their tones sharper. Bismuth and lead are also often used to modify the tone, which each metal affects differently. The addition of antimony and bismuth is frequently made by the founder to give a more crystalline grain to the alloy. All these conditions are, however, prejudicial to the sonorousness of bells, and of very doubtful utility. Rapid refrigeration increases the sonorousness of all these alloys. Hence M. D'Arcet recommends that the "pieces" be heated to a cherry-red after they are cast, and after having been suddenly plunged into cold water, that they be submitted to well-regulated pressure by skillful hammering, until they assume their proper form ; after which they are to be again heated and allowed to cool slowly in the air. This is the method adopted by the Chinese with their gongs, etc., a casing of sheet iron being employed by them to support and protect the pieces during the exposure to heat. In a general way, however, bells are formed and completed by simple casting. This is necessarily the case with all very large bells. Where the quality of their tones is the chief object sought after, the greatest care should be taken to use commercially pure copper. The presence of a very little lead or any similar metal greatly lessens the sonorousness of this alloy; while that of silver increases it.
The specific gravity of a large bell is seldom uniform through its whole substance; nor can the specific gravity from any given portion of its constituent metals be exactly calculated owing to the many interfering circumstances. The nearer this uniformity is approached, or, in other words, chemical combination is complete, the more durable and finer-toned will be the bell. In general, it is found necessary to take about one-tenth more metal than the weight of the intended bell, or bells, in order to allow for waste and scorification during the operations of fusing and casting.
BISMUTH ALLOYS.
Bismuth possesses the unusual quality of expanding in cooling. It is, therefore, introduced in many alloys to reduce or check shrinkage in the mold.
For delicate castings, and for taking impressions from dies, medals, etc., various bismuth alloys are in use, whose composition corresponds to the following figures:
I II III IV
Bismuth 6 5 2 8
Tin 3 2 1 3
Lead 13 3 1 5
V. Cliche Metal. This alloy is composed of tin, 48 parts; lead, 32.5; bismuth, 9; and antimony, 10.5. It is especially well adapted to dabbing rollers for printing cotton goods, and as it possesses a considerable degree of hardness, it wears well.
VI. For filling out defective places in metallic castings, an alloy of bismuth 1 part, antimony 3, lead 8, can be advantageously used.
VII. For Cementing Glass. Most of the cements in ordinary use are dissolved, or at least softened, by petroleum. An alloy of lead 3 parts, tin 2, bismuth 2.5, melting at 212º F., is not affected by petroleum, and is therefore very useful for cementing lamps made of metal and glass combined.
LIPOWITZ'S BISMUTH ALLOY:
See Cadmium Alloys.
BRASS.
In general brass is composed of two-thirds copper and one-third zinc, but a little lead or tin is sometimes advantageous, as the following:
I. Red copper, 66 parts; zinc, 34 parts; lead, 1 part.
II. Copper, 66 parts; zinc, 32 parts;
tin, 1 part; lead, 1 part.
III. Copper, 64.5 parts; zinc, 33.5 parts; lead, 1.5 parts; tin, 0.5 part.
Brass-Aluminum. A small addition of aluminum to brass (1.5 to 8 per cent) great-
[53]
ALLOYS
ly increases its hardness and elasticity, and this alloy is also easily worked for any purpose. Brass containing 8 per cent of aluminum has the valuable property of being but slightly affected by acids or gases.
A larger percentage of aluminum makes the brass brittle. It is to be noted that aluminum brass decreases very materially in volume in casting, and the casts must be copied slowly or they will be brittle. It is an alloy easily made, and its low price, combined with its excellent qualities, would seem to make it in many cases an advantageous substitute for the expensive phosphorous bronze.
Bristol Brass (Prince's Metal). This alloy, which possesses properties similar to those of French brass, is prepared in the following proportions:
I II III
Copper 75.7 67.2 60.8
Zinc 24.3 32.8 39.2
Particular care is required to prevent the zinc from evaporating during the fusing, and for this purpose it is customary to put only half of it into the first melting, and to add the remainder when the first mass is liquefied.
Brass-Iron (Aich's Metal). This is a variety of brass with an dmixture of iron, which gives it a considerable degree of tenacity. It is especially adapted for purposes which require a hard and, at the same time, tenacious metal. Analyses of the various kinds of this metal show
considerable variation in the proportions. Even the amount of iron, to which the hardening effect must be attributed, may vary within wide limits without materially modifying the tenacity which is the essential characteristic of this alloy.
I. The best variety of Aich's metal consists of copper, 60 parts; zinc, 38.2; iron, 1.8. The predominating quality of this alloy is its hardness, which is claimed to be not inferior to that of certain kinds of steel. It has a beautiful golden-yellow color, and is said not to oxidize easily, a valuable property for articles exposed to the action of air and water.
II. Copper, 60.2 parts; zinc, 38.2; iron, 1.6. The permissible variations in the content of iron are from 0.4 to 3 per cent.
Sterro metal may properly be considered in connection with Aich's metal, since its constituents are the same and its properties very similar. The principal difference between the two metals is that sterro metal contains a much larger amount of iron. The composition of this alloy varies considerably with different manufacturers.
III. Two varieties of excellent quality are the product of the Rosthorn factory, in Lower Austria copper, 55.33 parts; zinc, 41.80; iron, 4.66. Also
IV. English sterro metal (Gedge's alloy for ship sheathing), copper, 60 parts; zinc, 38.125; iron, 1.5.
The great value of this alloy lies in its strength, which is equaled only by that of the best steel. As an illustration of this, a wrought-iron pipe broke with a pressure of 267 atmospheres, while a similar pipe of sterro metal withstood the enormous pressure of 763 atmospheres without cracking. Besides its remarkable strength, it possesses a high degree of elasticity, and is, therefore, particularly suitable for purposes which require the combination of these two qualities, such as the construction of hydraulic cylinders. It is well known that these cylinders, at a certain pressure, begin to sweat, that is, the interior pressure is so great that the water permeates through the pores of the steel. With a sterro metal cylinder, the pressure can be considerably increased without any moisture being perceptible on the outside of the cylinder.
Sterro metal can be made even more hard and dense, if required for special purposes, but this is effected rather by mechanical manipulation than by any change in the chemical composition. If rolled or hammered in heat, its strength is increased, and it acquires, in addition, an exceedingly high degree of tenacity. Special care must be taken, however, in hammering not to overheat the metal, as in this case it would become brittle and might crack under the hammer. Sterro metal is especially suitable for all the purposes for which the so-called red metal has been in the past almost exclusively used. Axle bearings, for example, made of sterro metal have such excellent qualities that many machine factories are now using this material entirely for the purpose.
Cast Brass. The various articles of bronze, so called, statuettes, clock cases, etc., made in France, where this industry has attained great perfection and extensive proportions, are not, in many cases, genuine bronze, but fine cast brass. Following are the compositions of a few mixtures of metals most frequently used by French manufacturers:
Copper Zinc Tin Lead
1 63.70 33.55 2.50 0.25
II 64.45 32.44 0.25 2.86
III 70.90 24.05 2.00 3.05
IV 72.43 22.75 1.87 2.95
[54]
ALLOYS
Their special advantage is that they can be readily cast, worked with file and chisel, and easily gilded.
To Cast Yellow Brass. If good, clean, yellow brass sand castings are desired, the brass should not contain over 30 per cent of zinc. This will assure an alloy of good color and one which will run free and clean. Tin or lead may be added without affecting the property of casting clean. A mixture of 7 pounds of copper, 3 pounds of spelter, 4 ounces of tin, and 3 ounces of lead makes a good casting alloy and one which will cut free and is strong. If a stronger alloy be desired, more tin may be added, but 4 ounces is usually sufficient. If the alloy be too hard, reduce the proportion of tin.
Leaf Brass. This alloy is also called Dutch gold, or imitation gold leaf. It is made of copper, 77.75 to 84.5 parts; zinc, 15.5 to 22.25. Its color is pale or bright yellow or greenish, according to the proportions of the metals. It has an unusual degree of ductility.
Malleable Brass. This metal is affected less by sea water than pure copper, and was formerly much used for ship sheathing, and for making nails and rivets which were to come in contact with sea water. At the present day it has lost much of its importance, since all the larger ships are made of steel. It is usually composed of copper, 60 to 62 parts; and zinc, 40 to 38 parts. It is sometimes called yellow metal, or Müntz metal (called after its inventor), and is prepared with certain precautions, directed toward obtaining as fine a grain as possible, experience having shown that only a fine-grained alloy of uniform density can resist the action of the sea water evenly. A metal of uneven density will wear in holes. To obtain as uniform a grain as possible, small samples taken from the fused mass are cooled quickly and examined as to fracture. If they do not show the desired uniform grain, some zinc is added to the mass. After it has permeated the whole mass, a fresh sample is taken and tested, this being continued until the desired result is reached. It is scarcely necessary to remark that considerable experience is required to tell the correct composition of the alloy from the fracture. The mass is finally poured into molds and rolled cold. Malleable brass can be worked warm, like iron, being ductile in heat, a valuable quality.
Experiments with malleable brass show that all alloys containing up to 58.33 per cent of copper and up to 41.67 per cent of zinc are malleable. There is, in addition, a second group of such alloys, with 61.54 per cent of copper and 38.46 per cent of zinc, which are also malleable in heat.
The preparation of these alloys requires considerable experience, and is best accomplished by melting the metals together in the usual manner, and heating the fused mass as strongly as possible. It must be covered with a layer of charcoal dust to prevent oxidation of the zinc. The mass becomes thinly fluid, and an intimate mixture of the constituents is effected. Small pieces of the same alloy are thrown into the liquid mass until it no longer shows a reflecting surface, when it is cast into ingots in iron molds. The ingots are plunged into water while still red-hot, and acquire by this treatment a very high degree of ductility. The alloy, properly prepared, has a fibrous fracture and a reddish-yellow color.
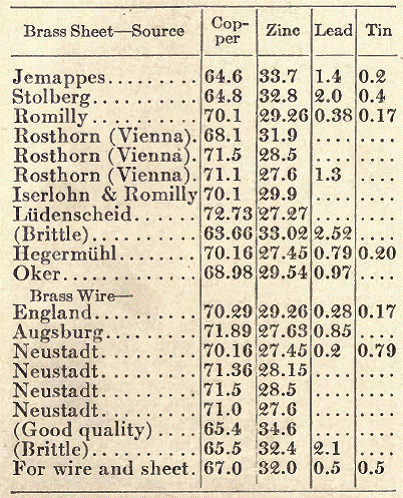
Sheet Brass (For Sheet and Wire). In the preparation of brass for the manufacture of wire, an especially pure quality of copper must be used; without this, all efforts to produce a suitable quality of brass will be in vain. That pure copper is indispensable to the manufacture of good, ductile brass may be seen from the great difference in the composition of the various kinds, all of which answer their purpose, but contain widely varying quantities of copper and zinc. The following table shows the composition of some excellent qualities of brass suitable for making sheet and wire:
[55}
ALLOYS
As the above figures show, the percentage of zinc in the different kinds of brass lies between 27 and 34. Recently, alloys containing a somewhat larger quantity of zinc have been used, it having been found that the toughness and ductility of the brass are increased thereby, without injury to its tenacity. Alloys containing up to 37 per cent of zinc possess a high degree of ductility in the cold, and are well adapted for wire and sheet.
Gilders' Sheet Brass. Copper, 1 part; zinc, 1 part; tin, 1/10 part; lead, 1/10 part. Very readily fusible and very dense.
White Brass. Birmingham platina is an alloy of a pure white, almost silver-white color, remaining unaffected by tolerably long exposure to the atmosphere. Unfortunately this alloy is so brittle that it can rarely be shaped except by casting. It is used only in the manufacture of buttons. The alloy is poured into molds giving rather sharp impressions and allowing the design on the button (letters or coat of arms) to stand out prominently with careful stamping. The composition of this alloy, also known by the name of platinum lead, is as follows:
I II
Copper 46.5 4
Zinc 53.5 16
III. Zinc, 80 parts; copper, 10 parts; iron, 10 parts.
BRITANNIA METAL.
Britannia metal is an alloy consisting principally of tin and antimony. Many varieties contain only these two metals, and may be considered simply as tin hardened with antimony, while others contain, in addition, certain quantities of copper, sometimes lead, and occasionally, though rarely on account of its cost, bismuth. Britannia metal is always of a silvery-white color, with a bluish tinge, and its hardness makes it capable of taking a high polish, which is not lost through exposure to the air. Ninety per cent of tin and 10 per cent of antimony gives a composition which is the best for many purposes, especially for casting, as it fills out the molds well, and is readily fusible. In some cases, where articles made from it are to be subjected to constant wear, a harder alloy is required. In the proportions given above, the metal is indeed much harder than tin, but would still soon give way under usage.
A table is appended, giving the composition of some of the varieties of Britannia metal and their special names.
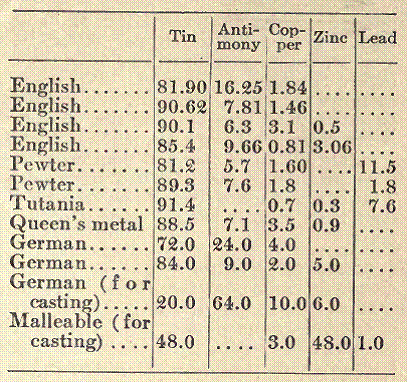
Britannia metal is prepared by melting the copper alone first, then adding a part of the tin and the whole of the antimony. The heat can then be quickly moderated, as the melting point of the new alloy is much lower than that of copper. Finally, the rest of the tin is added, and the mixture stirred constantly for some time to make it thoroughly homogeneous.
An alloy which bears a resemblance to Britannia metal is Ashberry metal, for which there are two formulas.
I II
Copper 2 3
Tin. 8 79
Antimony 14 15
Zinc 1 2
Nickel 2 1
BRONZES.
The composition of bronze must be effected immediately before the casting, for bronze cannot be kept in store ready prepared. In forming the alloy, the refactory compound, copper, is first melted separately, the other metals, tin, zinc, etc., previously heated, being then added; the whole is then stirred and the casting carried out without loss of time. The process of forming the alloy must be effected quickly, so that there may be no loss of zinc, tin, or lead through oxidation, and also no interruption to the flow of metal, as metal added after an interval of time will not combine perfectly with the metal already poured in.
It is important, therefore, to ascertain the specific weights of the metals, for the heavier metal will naturally tend to sink to the bottom and the lighter to collect at the top. Only in this way, and by vigorous stirring, can the complete blending of the two metals be secured. In adding the zinc, great care
[56]
ALLOYS
must be taken that the latter sinks at once to the level of the copper, otherwise a considerable portion will be volatilized before reaching the copper. When the castings are made, they must be cooled as quickly as possible, for the components of bronze have a tendency to form separate alloys of various composition, thus producing the so-called tin spots. This is much more likely to occur with a slow than with a sudden cooling of the mass.
Annealing Bronze. This process is more particularly employed in the preparation of alloys used in the manufacture of cymbals, gongs, bells, etc. The alloy is naturally brittle, and acquires the properties essential to the purpose for which it is intended only after casting.
The instruments are plunged into cold water while red-hot, hammered, reheated, and slowly cooled, when they become soft and sonorous. The alloy of copper and tin has the peculiar property that, whereas steel becomes hard through cooling, this mixture, when cooled suddenly, becomes noticeably soft and more malleable. The alloy is heated to a dark-red heat, or, in the case of thin articles, to the melting point of lead, and then plunged in cold water. The alloy may be hammered without splitting or breaking.
Aluminum Bronze. This is prepared by melting the finest copper in a crucible, and adding the aluminum. The copper is cooled thereby to the thickly fluid point, but at the moment of the combination of the two metals, so much heat is released that the alloy becomes white hot and thinly fluid. Aluminum bronze thus prepared is usually brittle, and acquires its best qualities only after having been remelted several times. It may be remarked that, in order to obtain a bronze of the best quality, only the very purest copper must be used; with an inferior quality of copper, all labor is wasted. Aluminum bronze is not affected by exposure to the air; and its beautiful color makes it very suitable for manufacturing various ornamental articles, including clock cases, door knobs, etc.
Aluminum bronze wire is almost as strong as good steel wire, and castings made from it are almost as hard as steely iron; its resistance to bending or sagging is great.
I. A good formula is 90 to 95 per cent of aluminum and 5 to 10 per cent of copper, of golden color, which keeps well in the air, without soon becoming dull and changing color like pure copper and its alloys with tin and zinc (bronze, brass, etc.). It can be cast excellently, can be filed well and turned, possesses an extraordinary hardness and firmness, and attains a high degree of polish; it is malleable and forgeable. On the latter quality are founded applications which were formerly never thought of, viz.: forged works of art for decorative purposes. An alloy of 95 parts aluminum and 5 parts copper is used here. The technical working of bronze is not materially different from that of iron. The metal, especially in a hot condition, is worked like iron on the anvil, with hammer and chisel, only that the temperature to be maintained in forging lies between dark and light cherry red. If the articles are not forged in one piece and the putting together of the separate parts becomes necessary, riveting or soldering has to be resorted to. Besides forging, aluminum bronze is well suited for embossing, which is not surprising considering the high percentage of copper. After finishing the pieces, the metal can be toned in manifold ways by treatment with acid.
II. Copper, 89 to 98 per cent; aluminum and nickel, 1 to 2 per cent. Aluminum and nickel change in the opposite way, that is to say, in increasing the percentage of nickel the amount of aluminum is decreased by the equal quantity. It should be borne in mind that the best ratio is aluminum, 9.5 per cent; nickel, 1 to 1.5 per cent at most. In preparing the alloy a deoxidizing agent is added, viz., phosphorus to 0.5 per cent; magnesium to 1.5 per cent. The phosphorus should always be added in the form of phosphorous copper or phosphor aluminum of exactly determined percentage. It is first added to the copper, then the aluminum and the nickel, and finally the magnesium, the last named at the moment of liquidity, are admixed.
III. A gold bronze, containing 3 to 5 per cent aluminum; specific gravity. 8.37 to 8.15. Handsome golden color. This alloy oxidizes less on heating than copper and iron, and is therefore especially adapted for locomotive fireboxes and spindles, etc.
IV. A steel bronze containing on an average 8.5 per cent aluminum including 1 per cent silicium); specific gravity, 7.7. Very ductile and tough, but slightly elastic; hence its use is excluded where, with large demands upon tension and pressure, no permanent change of form must ensue. This is changed by working, such as rolling, drawing, etc. Es-
[57]
ALLOYS
pecially useful where infrangibility is desired, as in machinery, ordnance, etc. At high temperature this bronze loses its elasticity again.
V. This contains 8.5 per cent aluminum and 1 A to 2 per cent silicium. Its use is advisable in cases where the metal is to possess a good elasticity, even in the cast state, and to retain it after being worked in red heat.
VI. An acid bronze, containing 10 per cent aluminum; specific gravity,
7.65. Especially serviceable to resist oxidation and the action of acids.
VII. Diamond bronze, containing 10 per cent aluminum and 2 per cent
silicium. Specific gravity, 7.3. Very hard; of great firmness, but brittle.
Art Bronzes. (See also Aluminum Bronzes and Japanese Bronzes under
this title.)
I. Copper, 84 parts; zinc, 11 parts; tin, 5 parts.
II. Copper, 90 parts; zinc, 6 parts; tin, 2 parts; lead, 2 parts.
III. Copper, 65 parts; zinc, 30 parts; tin, 5 parts.
IV. Copper, 90 parts; tin, 5 parts; zinc, 4 parts; lead, 1 part.
V. Copper, 85 parts; zinc, 10 parts; tin, 3 parts; lead, 2 parts.
VI. Copper, 72 parts; zinc, 23 parts; tin, 3 parts; lead, 2 parts.
Statuary Bronze. Many of the antique statues were made of genuine bronze, which has advantages for this purpose, but has been superseded in modern times by mixtures of metals containing, besides copper and tin the constituents of real bronze a quantity of zinc, the alloy thus formed being really an intermediate product between bronze and brass. The reason for the use of such mixtures lies partly in the comparative cheapness of their production as compared with genuine bronze, and partly in the purpose for which the metal is to be used. A thoroughly good statuary bronze must become thinly fluid in fusing, fill the molds out sharply, allow of being easily worked with the file, and must take on the beautiful green coating called patina, after being exposed to the air for a short time.
Genuine bronze, however strongly heated, does not become thin enough to fill out the molds well, and it is also difficult to obtain homogeneous castings from it. Brass alone is also too thickly fluid, and not hard enough for the required fine chiseling or chasing of the finished object. Alloys containing zinc and tin, in addition to copper, can be prepared in such a manner that they will become very thinly fluid, and will give fine castings which can easily be worked with the file and chisel. The best proportions seem to be from 10 to 18 per cent of zinc and from 2 to 4 per cent of tin. In point of hardness, statuary bronze holds an intermediate position between genuine bronze and brass, being harder and tougher than the latter, but not so much so as the former.
Since statuary bronze is used principally for artistic purposes, much depends upon the color. This can be varied from pale yellow to orange yellow by slightly varying the content of tin or zinc, which must, of course, still be kept between the limits given above. Too much tin makes the alloy brittle and difficult to chisel; with too much zinc, on the other hand, the warm tone of color is lost, and the bronze does not acquire a fine patina.
The best proportions for statuary bronze are very definitely known at the present day; yet it sometimes happens that large castings have not the right character. They are either defective in color, or they dc not take on a fine patina, or they are difficult to chisel. These phenomena may be due to the use of impure metals containing oxides, iron, lead, etc. or to improper treatment of the alloy in melting. With the most careful work possible, there is a considerable loss in melting 3 per cent at the very least, and. sometimes as, much as 10. This is due to the large proportion of zinc, and it is evident that, in consequence of it, the nature of the alloy will be different from what might be expected from the quantities of metals used in its manufacture.
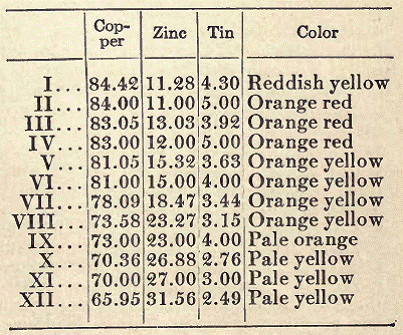
It has been remarked that slight variations in composition quickly change the color of the alloy. The following table gives a series of alloys of different colors, suitable for statuary bronze:
[58]
ALLOYS
Perhaps the most satisfactory bronze metal is the alloy used in France for more than a century. It contains 91.60 per cent of copper, 5.33 per cent of zinc, 1.70 per cent of tin, and 1.37 per cent of lead.
Somewhat more zinc is taken for articles to be gilded.
Bismuth Bronze. Copper, 52 parts; nickel, 30 parts; zinc, 12 parts; lead, 5 parts; bismuth, 1 part. For metallic mirrors, lamp reflectors, etc.
Gun Bronze. See Phosphor Bronze under this title.
Japanese Bronzes. The formulas given below contain a large percentage of lead, which greatly improves the patina. The ingredients and the ratio of their parts for several sorts of modern Japanese bronze follow:
I. Copper, 81.62 per cent; tin, 4.61 per cent; lead, 10.21 per cent.
II. Copper, 76.60 per cent; tin, 4.38 per cent; lead, 11.88 per cent; zinc, 6.53 per cent.
III. Copper, 88.55 per cent; tin, 2.42 per cent; lead, 4.72 per cent; zinc, 3.20 per cent.
Sometimes a little antimony is added just before casting, and such a composition would be represented more nearly by this formula:
IV. Copper, 68.25 per cent; tin, 5.47 per cent; zinc, 8.88 per cent; lead, 17.06 per cent; antimony, 0.34 per cent.
For imitation Japanese bronze, see Plating under Bronzing.
Machine Bronze.
I. Copper, 89 per cent; tin, 11 per cent.
II. Copper, 80 per cent; tin, 16 per cent.
Phosphor Bronze. Phosphor bronze is bronze containing varying amounts of phosphorus, from a few hundredths of 1 per cent to 1 or 2 per cent. Bronze containing simply copper and tin is very liable to be defective from the presence of oxygen, sulphur, or occluded gases. Oxvgen causes the metal to be spongy and weak. Sulphur and occluded gases cause porosity. Oxygen gets into the metal by absorption from the air. It can be eliminated by adding to the metal something which combines with the oxygen and then fluxes off. Such deoxidizers are zinc, antimony, aluminum, manganese, silicon, and phosphorus. Sulphur and occluded gases can be eliminated by melting the metal, exposing it to the air, and letting it thus absorb some oxygen, which then burns the sulphur and gas. The oxygen can then be removed by adding one of the above-mentioned deoxidizers. The important use of phosphorus in bronze is, therefore, to remove oxygen and also indirectly to destroy occluded gas and sulphur.
A bronze is sometimes made with an extra high percentage of phosphorus, namely, 6 per cent. This alloy is made so as to have phosphorus in convenient form for use, and the process of manufacture is as follows: Ninety pounds of copper are melted under charcoal in a No. 70 crucible, which holds about 200 pounds of metal when full; 11 pounds of tin are added and the metal is allowed to become hot. The crucible is then removed from the furnace and 7 pounds of phosphorus are introduced in the following manner: A 3-gallon stone jar, half full of dilute solution of blue vitriol, is weighed. Then the weights are increased 7 pounds, and phosphorus in sticks about 4 inches long is added till the scales balance again. The phosphorus is left in this solution half an hour or longer, the phosphorus being given a coating of copper, so that it may be dried and exposed to the air without igniting. Have ready a pan about 30 inches square and 6 inches deep, containing about 2 inches of water. Over the water is a wire netting, which is laid loose on ledges or supports along the inner sides of the pan. On the netting is blotting paper, and on this the phosphorus is laid to dry when taken out of the blue-vitriol solution. The pan also has a lid which can be put down in case of ignition of the phosphorus.
The phosphorus is now ready for introduction into the metal. This is done by means of a cup-shaped instrument called a retort or phosphorizer. One man holds the retort on the rim of the crucible in a horizontal position. A second man takes about three pieces of phosphorus and throws them into the retort. The first man then immediately plunges the mouth of the retort below the surface of the metal before the phosphorus has a chance to fall or flow out. Of course the phosphorus immediately melts and also begins to volatilize. As the phosphorus comes in contact with the metal, it combines with it. This process is continued till all the 7 pounds of phosphorus has been put into the metal. The metal is then poured into slabs about 3 inches by 4 inches by 1 inch thick. The metal is so hard that a greater thickness would make it difficult to break it up. When finished, the metal contains, by analysis, 6 per cent of phosphorus. When phosphorus is to be added to metal, a little of this hardener is employed.
Copper is a soft, ductile metal, with its melting point at about 2,000º F. Mol-
[59]
ALLOYS
ten copper has the marked property of absorbing various gases. It is for this reason that it is so difficult to make sound castings of clear copper. Molten copper combines readily with the oxygen of the air, forming oxide of copper, which dissolves in the copper and mixes homogeneously with it.
A casting made from such metal would be very spongy. The bad effect of oxygen is intended to be overcome by adding zinc to the extent of 1 per cent or more. This result can be much more effectively attained by the use of aluminum, manganese, or phosphorus. The action of these substances is to combine with the oxygen, and as the product formed separates and goes to the surface, the metal is left in a sound condition. Aluminum and manganese deoxidize copper and bronze very effectively, and the oxide formed goes to the surface as a scum. When a casting is made from such metal, the oxide or scum, instead of freeing itself from the casting perfectly, generally remains in the top part of the casting mixed with the metal, as a fractured surface will show. Phosphorus deoxidizes copper, and the oxide formed leaves the metal in the form of a gas, so that a casting made from such metal shows a clean fracture throughout, although the metal is not so dense as when aluminum or manganese is used.
Copper also has the property of absorbing or occluding carbon monoxide. But the carbonic oxide thus absorbed is in a different condition from the oxygen absorbed. When oxygen is absorbed by copper, the oxygen combines chemically with the copper and loses its own identity as a gas. But when coal gas is absorbed by the copper, it keeps its own physical identity and simply exists in the copper in a state of solution. All natural waters, such as lake water, river water, spring water, etc., contain air in solution or occlusion. When such water is cooled and frozen, just at the time of changing from the liquid to the solid state, the dissolved gas separates and forms air bubbles, which remain entangled in the ice. The carbonic oxide which is dissolved or occluded in copper acts in exactly the same way.
Hydrogen acts in exactly the same manner as carbonic oxide. Sulphur also has a bad effect upon copper and bronze. Sulphur combines with copper and other metals, forming sulphide of copper, etc. When molten copper or bronze containing sulphur comes in contact with air it absorbs some oxygen, and this in turn ombines with the sulphur present, forming sulphur dioxide, which is a gas which remains occluded in the metal.
Tin is a soft, white metal, melting at 440º F. Toward gases it acts something like copper, but not in so marked a degree. Although copper and tin are both soft, vet when mixed they make a harder metal. When bronze cools from the molten state, the copper and the copper-tin alloy tend to crystallize by themselves. The quicker the cooling occurs the less separation will there be, and also the fracture will be more homogeneous in appearance.
Gun bronze contains copper and tin in the proportion of 9 or 10 parts of copper to 1 of tin. This is the metal used when an ordinary bronze casting is wanted. A harder bronze is copper and tin in the ratio of 6 to 1. This is often used as a bearing metal. When either of these metals is to be turned in the machine shop, they should contain about 3 per cent of lead, which will make them work very much better, but it also decreases their tensile strength. Bearing metal now generally contains about 10 per cent of lead, with copper and tin in varying ratios. The large percentage of lead is put in that the metal may wear away slower. Lead, although a metal having properties similar to tin, acts entirely different toward copper. Copper and tin have a good deal of affinity for each other, but copper and lead show no attraction at all for each other. Copper and tin mix in all proportions, but copper and lead mix only to a very limited extent. About 3 per cent of lead can be mixed with copper. With bronze about 15 per cent to 20 per cent of lead can be mixed. In bearing bronze the lead keeps its own physical properties, so that the constituent lead melts long before the metal attains a red heat. It sometimes happens when a bearing runs warm that the lead actually sweats out and forms pimples on the metal. Or, sometimes, in remelting a bearing bronze casting the lead may be seen to drop out while the metal is warming up. All of these metals, however, should contain something to flux or deoxidize them, such as zinc, manganese, aluminum, silicon, antimony, or phosphorus.
The phosphor bronze bearing metal in vogue has the following composition: Coppper, 79.7 per cent; tin, 10 per cent; lead, 10 percent; and phosphorus, 0.3 per cent.
Melt 140 pounds of copper in a No. 70 pot, covering with charcoal. When copper is all melted, add 17 1/2 pounds of tin to 17 1/2 pounds of lead, and allow the metal to become sufficiently warm, but
[60]
ALLOYS
not any hotter than is needed. Then add 10 pounds of "hardener" (made as previously described) and stir well. Remove from furnace, skim off the charcoal, cool the metal with gates to as low a temperature as is consistent with getting a good casting, stir well again, and pour. The molds for this kind of work are faced with plumbago.
There are several firms that make phosphor-bronze bearings with a composition similar to the above one, and most of them, or perhaps all, make it by melting the metals and then charging with phosphorus to the extent of 0.7 to 1 per cent. But some metal from all brands contains occluded gas. So that after such metal is cast (in about two minutes or so) the metal will ooze or sweat out through the gate, and such a casting will be found to be porous. But not one such experience with metal made as described above has yet been found.
This practical point should be heeded, viz., that pig phosphor bronze should be brought to the specifications that the metal should have shrunk in the ingot mold in cooling, as shown by the concave surface of the upper side, and that it should make a casting in a sand mold without rising in the gate after being poured.
In bearing metal, occluded gas is very objectionable, because the gas, in trying to free itself, shoves the very hard copper-tin compound (which has a low melting point and remains liquid after the copper has begun to set) into spots, and thus causes hard spots in the metal.
Phosphorus is very dangerous to handle, and there is great risk from fire with it, so that many would not care to handle the phosphorus itself. But phosphor copper containing 5 per cent of phosphorus, and phosphor tin containing 2 to 7 per cent of phosphorus, and several other such alloys can be obtained in the market. It may be suggested to those who wish to make phosphor bronze, but do not want to handle phosphorus itself, to make it by using the proper amounts of one of these high phosphorus alloys. In using phosphorus it is only necessary to use enough to thoroughly deoxidize the metal, say 0.3 per cent. More than this will make the metal harder, but not any sounder.
Phosphor bronze is not a special kind of alloy, but any bronze can be made into phosphor bronze; it is, in fact, simply a deoxidized bronze, produced under treatment with phosphorus compounds.
Although the effect of phosphorus in improving the quality of bronze has been known for more than fifty years, it is only of late that the mode for preparing phosphor bronze has been perfected. It is now manufactured in many localities. Besides its action in reducing the oxides dissolved in the alloy, the phosphorus exerts another very material influence upon the properties of the bronze. The ordinary bronzes consist of mixtures in which the copper is really the only crystallized constituent, since the tin crystallizes with great difficulty. As a consequence of this dissimilarity in the nature of the two metals, the alloy is not so solid as it would be if both were crystallized. The phosphorus causes the tin to crystallize, and the result is a more homogeneous mixture of the two metals.
If enough phosphorus is added, so that its presence can be detected in the finished bronze, the latter may be considered an alloy of crystallized phosphor tin with copper. If the content of phosphor is still more increased, a part of the copper combines with the phosphorus, and the bronze then contains, besides copper and tin, compounds of crystallized copper phosphide with phosphide of tin. The strength and tenacity of the bronze are not lessened by a larger amount of phosphorus, and its hardness is considerably increased. Most phosphor bronzes are equal in this respect to the best steel, and some even surpass it in general properties.
The phosphorus is added to the bronze in the form of copper phosphide or phosphide of tin, the two being sometimes used together. They must be specially prepared for this purpose, and the best methods will be here given. Copper phosphide is prepared by heating a mixture of 4 parts of superphosphate of lime, 2 parts of granulated copper, and 1 part of finely pulverized coal in a crucible at a temperature not too high. The melted copper phosphide, containing 14 per cent of phosphorus, separates on the bottom of the crucible.
Tin phosphide is prepared as follows: Place a bar of zinc in an aqueous solution of tin chloride. The tin will be separated in the form of a sponge-like mass. Collect it, and put it into a crucible, upon the bottom of which sticks of phosphorus have been placed. Press the tin tightly into the crucible, and expose to a gentle heat. Continue the heating until flames of burning phosphorus are no longer observed on the crucible. The pure tin phosphide, in the form of a coarsely crystalline mass, tin-white in color, will be found on the bottom of the crucible.
To prepare the phosphor bronze, the
[61]
ALLOYS
alloy to be treated is melted in the usual way, and small pieces of the copper phosphide and tin phosphide are added.
Phosphor bronze, properly prepared, has nearly the same melting point as that of ordinary bronze. In cooling, however, it has the peculiarity of passing directly from the liquid to the solid state, without first becoming thickly fluid. In a melted state it retains a perfectly bright surface, while ordinary bronze in this condition is always covered with a thin film of oxide.
If phosphor bronze is kept for a long time at the melting point, there is not any loss of tin, but the amount of phosphorus is slightly diminished.
The most valuable properties of phosphor bronze are its extraordinary tenacity and strength. It can be rolled, hammered, and stretched cold, and its strength is nearly double that of the best ordinary bronze. It is principally used in cases where great strength and power of resistance to outward influences are required, as, for instance, in objects which are to be exposed to the action of sea water.
Phosphor bronze containing about 4 per cent of tin is excellently well adapted for sheet bronze. With not more than 5 per cent of tin, it can be used, forged, for firearms. Seven to 10 per cent of tin gives the greatest hardness, and such bronze is especially suited to the manufacture of axle bearings, cylinders for steam fire engines, cogwheels, and, in general, for parts of machines where great strength and hardness are required. Phosphor bronze, if exposed to the air, soon becomes covered with a beautiful, closely adhering patina, and is therefore well adapted to purposes of art. The amount of phosphorus added varies from 0.25 to 2.5 per cent, according to the purpose of the bronze. The composition of a number of kinds of phosphor bronze is given below:
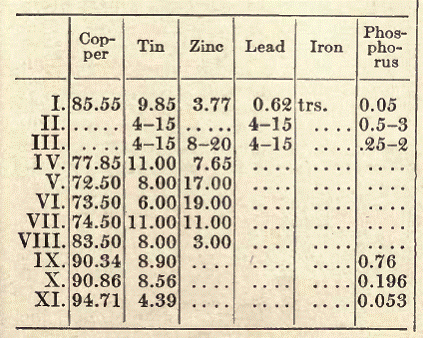
I for axle bearings, II and III for harder and softer axle bearings, IV to VIII for railroad purposes, IV especially for valves of locomotives, V and VI axle bearings for wagons, VII for connecting rods, VIII for piston rods in hydraulic presses.
Steel Bronze. Copper, 60; ferromanganese (containing 70 to 80 per cent manganese), 40; zinc, 15.
Silicon Bronze. Silicon, similarly to phosphorus, acts as a deoxidizing agent, and the bronzes produced under its influence are very ductile and elastic, do not rust, and are very strong. On account of these qualities silicon bronze is much used for telegraph and telephone wires. The process of manufacture is similar to that of phosphor bronze; the silicon is used in the form of copper silicide. Some good silicon bronzes are as follows:
I II
Copper 97.12 97.37
Tin 1.14 1.32
Zinc l.10 1.27
Silicon 0.05 0.07
Sun Bronze. The alloy called sun bronze contains 10 parts of aluminum,
30 to 50 parts of copper, and 40 to 60 parts of cobalt. The mixture known by the name of metalline has 25 per cent of aluminum, 30 of copper, 10 of iron, and 35 of cobalt. These alloys melt at a point approaching the melting point of copper, are tenacious, ductile, and very hard.
Tobin Bronze. This alloy is nearly similar in composition and properties to Delta metal.

The alloy marked IV is sometimes called deoxidized bronze.
Violet-colored bronze is 50 parts copper and 50 parts antimony.
CADMIUM ALLOYS:
See also Fusible Alloys.
Lipowitz's Alloy. I. This alloy is composed of cadmium, 3 parts; tin, 4; bismuth, 15; and lead, 8. The simplest method of preparation is to heat the metals, in small pieces, in a crucible, stirring constantly, as soon as fusion
[62]
ALLOYS
begins, with a stick of hard wood. The stirring is important, in order to prevent the metals, whose specific gravity varies considerably, from being deposited in layers. The alloy softens at 140º F. and melts completely at 158 P. The color is silvery white, with a luster like polished silver, and the metal can be bent, hammered, and turned. These properties would make it valuable for many purposes where a beautiful appearance is of special importance, but on account of the considerable amount of cadmium and bismuth which it contains, it is rather expensive, and therefore limited in use. Casts of small animals, insects, lizards, etc., have been prepared from it, which were equal in sharpness to the best galvanoplastic work. Plaster of Paris is poured over the animal to be cast, and after sharp drying, the animal is removed and the mold filled up with Lipowitz's metal. The mold is placed in a vessel of water, and by heating to the boiling point the metal is melted and deposited in the finest impressions of the mold.
This alloy is most excellent for soldering tin, lead, Britannia metal, and nickel, being especially adapted to the last two metals on account of its silver-white color. But here again its costliness prevents its general use, and cheaper alloys possessing the same properties have been sought. In cases where the silver-white color and the low melting point are not of the first importance, the alloys given below may very well be used in the place of it.
II. Cadmium alloy (melting point, 170 F.): Cadmium, 2 parts; tin, 3; lead, 11; bismuth, 16.
III. Cadmium alloy (melting point, 167º F.): Cadmium, 10 parts; tin, 3; lead, 8; bismuth, 8.
Cadmium alloys (melting point, 203º F.):
IV V VI
Cadmium 1 1 1 parts
Tin 2 3 1 "
Bismuth 3 5 2 "
VII. A very fusible alloy, melting at 150º F., is composed of tin, 1 or 2 parts; lead, 2 or 3; bismuth, 4 or 15; cadmium, 1 or 2.
VIII. Wood's alloy melts between 140 and 161.5º F. It is composed of lead, 4 parts; tin, 2; bismuth, 5 to 8; cadmium, 1 to 2. In color it resembles platinum, and is malleable to a certain extent.
IX. Cadmium alloy (melting point, 179.5º F.): Cadmium, 1 part; lead, 6 parts; bismuth, 7. This, like the preceding, can be used for soldering in hot water.
X. Cadmium alloy (melting point, 300º F.): 8 Cadmium, 2 parts; tin, 4; lead, 2. This is an excellent soft solder, with a melting point about 86 degrees below that of lead and tin alone.
Cadmium Alloys with Gold, Silver, and Copper.
I. Gold, 750 parts; silver, 166 parts; cadmium, 84 parts. A malleable and ductile alloy of green color.
II. Gold, 750 parts; silver, 125 parts; and cadmium, 125 parts. Malleable and ductile alloy of yellowish-green hue.
III. Gold, 746 parts; silver, 114 parts; copper, 97 parts; and cadmium, 43 parts. Likewise a malleable and ductile alloy of a peculiar green shade. All these alloys are suitable for plating. As regards their production, each must be carefully melted together from its ingredients in a covered crucible lined with coal dust, or in a graphite crucible.
Next, the alloy has to be remelted in a graphite crucible with charcoal (or rosin powder) and borax. If, in spite thereof, a considerable portion of the cadmium should have evaporated, the alloy must be refused once more with an addition of cadmium.
ALLOYS FOR CASTING COINS, MEDALLIONS, ETC.
Alloys which fulfill the requirements of the medalist, and capable, therefore, of reproducing all details, are the following:
I II
Tin 3 6 parts
Lead 13 8 "
Bismuth 6 14 "
III. A soft alloy suitable to take impressions of woodcuts, coins, metals, engravings, etc., and which must melt at a low degree of heat, is made out of bismuth, 3 parts; tin, l ½ parts; lead, 2 1/2 parts; and worn-out type, 1 part.
Acid-proof Alloy. This alloy is characterized by its power, of resisting the action of acids, and is therefore especially adapted to making cocks, pipes, etc., which are to come in contact with acid fluids. It is composed of copper, zinc, lead, tin, iron, nickel, cobalt, and antimony, in the following proportions:
Copper 74.75 parts
Zinc 0.61 "
Lead 16.35 "
Tin 0.91 "
Iron 0.43 "
Nickel | 24 " [parts each]
Cobalt |
Antimony 6.78 "
[63]
ALLOYS
Albata Metal. Copper, 40 parts; zinc, 32 parts; and nickel, 8 parts.
Alfenide Metal. Copper, 60 parts; zinc, 30; nickel, 10; traces of iron.
Bath Metal. This" alloy is used especially in England for the manufacture of teapots, and is very popular owing to the fine white color it possesses. It takes a high polish, and articles made from this alloy acquire in the course of time, upon only being rubbed with a white cloth, a permanent silver luster. The composition of Bath metal is copper, 55 parts; zinc, 45 parts.
Baudoin Metal. This is composed of 72 parts of copper, 16.6 of nickel, 1.8 of cobalt, 1 of zinc; 1/2 per cent of aluminum may be added.
CASTING COPPER:
Macht's Yellow Metal.
I. This alloy consists of 33 parts of copper and 25 of zinc. It has a dark golden-yellow color, great tenacity, and can be forged at a red heat, properties which make it especially suitable for fine castings.
II. Yellow. Copper, 67 to 70 parts; zinc, 33 to 30 parts.
III. Red. Copper, 82 parts; zinc, 18 parts.
Copper Arsenic. Arsenic imparts to copper a very fine white color, and makes it very hard and brittle. Before German silver was known, these alloys were sometimes used for the manufacture of such cast articles as were not to come in contact with iron. When exposed to the air, they soon lose their whiteness and take on a brownish shade. On account of this, as well as the poisonous character of the arsenic, they are very little used at the present time. Alloys of copper and arsenic are best prepared by pressing firmly into a crucible a mixture of 70 parts of copper and 30 of arsenic (the copper to be used in the form of fine shavings) and fusing this mixture in a furnace with a good draught, under a cover of glass.
Copper Iron. The alloys of copper and iron are little used in the industries of the present day, but it would seem that in earlier times they were frequently prepared for the purpose of giving a considerable degree of hardness to copper; for in antique casts, consisting principally of copper, we regularly find large quantities of iron, which leads to the supposition that they were added intention-
ally.
These alloys, when of a certain composition, have considerable strength and hardness. With an increase in the quantity of the iron the hardness increases, but the solidity is lessened. A copper and iron alloy of considerable strength, and at .the same time very hard, is made of copper, 66 parts; iron, 34. These alloys acquire, on exposure to air, an ugly color inclining toward black, and are therefore not adapted for articles of art.
Copper Nickel. A. Morrell, of New York, has obtained a patent on a nickel-copper alloy which he claims is valuable on account of its noncorrosive qualities, therefore making it desirable for ships, boiler tubes, and other uses where the metal comes much in contact with water. The process of making the metal is by smelting ore containing sulphide of nickel and copper, and besemerizing the resultant matter. This is
calcined in order to obtain the nickel and copper in the form of oxides. The latter are reduced in reverberating furnace with carbon, or the like, so as to produce an alloy which preferably contains 2 parts of nickel and 1 part of copper.
Delta Metal. An alloy widely used for making parts of machinery, and also for artistic purposes, is the so-called Delta metal. This is a variety of brass hardened with iron; some manufacturers add small quantities of tin and lead; also, in some cases, nickel. The following analysis of Delta metal (from the factory at Dusseldorf) will show its usual composition:
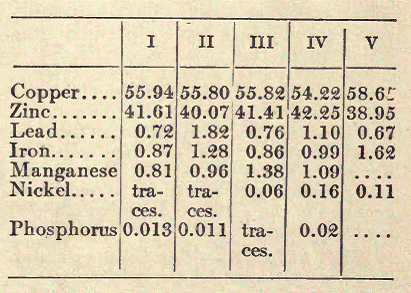
I is cast, II hammered, III rolled, and IV hot-stamped metal. Delta metal is produced by heating zinc very strongly in crucibles (to about 1600º F.), and adding ferromanganese or "spiegeleisen," producing an alloy of 95 per cent zinc and 5 per cent of iron. Copper and brass and a very small amount of copper phosphate are also added.
[64]
ALLOYS
Gong Metal. A sonorous metal for cymbals, gongs, and tam-tams consists of 100 parts of copper with 25 parts tin. Ignite the piece after it is cast and plunge it into cold water immediately.
Production of Minargent. This alloy consists of copper, 500 parts; nickel, 350; tungsten, 25, and aluminum, 5. The metal obtained possesses a handsome white color and greatly resembles silver.
Minofor. The so-called Minofor metal is composed of copper, tin, antimony, zinc, and iron in the following proportions:
I II
Copper 3.26 4
Tin 67.53 66
Antimony 17.00 20
Zinc 8.94 9
Iron 1
Minargent and Minofor are sometimes used in England for purposes in which the ordinary Britannia metal, 2 parts tin and 1 part antimony, might equally well be employed; the latter surpasses both of them in beauty of color, but they are, on the other hand, harder.
Retz Alloy. This alloy, which resists the corrosive action of alkalies and acids, is composed of 15 parts of copper, 2.34 of tin, 1.82 of lead, and 1 of antimony. It can be utilized in the manufacture of receivers, for which porcelain and ebonite are usually employed.
Ruoltz Metal. This comprises 20 parts of silver, 50 of copper, 30 of nickel. These proportions may, however, vary.
Tissier's Metal. This alloy contains arsenic, is of a beautiful tombac red color, and very hard. Its composition varies a great deal, but the peculiar alloy which gives the name is composed of copper, 97 parts; zinc, 2 parts; arsenic, 1 or 2. It may be considered a brass with a very high percentage of copper, and hardened by the addition of arsenic. It is sometimes used for axle bearings, but other alloys are equally suitable for this purpose, and are to be preferred on account of the absence of arsenic, which is always dangerous.
FILE ALLOYS. Many copper-tin alloys are employed for the making of files which, in distinction from the steel files, are designated composition files. Such alloys have the following compositions:
Geneva Composition Files.
I II
Copper 64.4 62
Tin 18.0 20
Zinc 10.0 10
Lead 7.6 8
Vogel’s Composition Files.
III IV V
Copper 57.0 61.5 73.0
Tin 28.5 31.0 19.0
Zinc 78.0 .... 8.0
Lead 7.0 8.5 8.0
VI. Another alloy for composition files is copper, 8 parts; tin, 2; zinc, l, and lead, 1 fused under a cover of borax.
EASILY FUSIBLE OR PLASTIC ALLOYS.
(These have a fusing point usually below 300º F.)
(See also Solders.)
I. Rose's Alloy. Bismuth, 2 parts; lead, 1 part; tin, 1 part. Melting point, 200º F.
II. Darcet Alloy. This is composed of 8 parts of bismuth, 5 of lead, and 3 of tin. It melts at 176º F. To impart greater fusibility, 1/16 part of mercury is added; the fusing is then lowered to 149º F.
III. Newton alloy melts at 212º F., and is composed of 5 parts of bismuth, 2 of lead, and 3 of tin.
IV. Wood's Metal.
Tin 2 parts
Lead 4 parts
Bismuth 5 to 8 parts
This silvery, fine-grained alloy fuses between 151 and 162º F., and is excellently adapted to soldering.
V. Bismuth, 7 parts; lead, 6 parts; cadmium, 1 part. Melting point, 180º F.
VI. Bismuth, 7 to 8 parts; lead, 4; tin, 2; cadmium, 1 to 2. Melting point, 149 to 160º F.
Other easily fusible alloys:
VII VIII IX
Lead 1 2 3
Tin 1 2 3
Bismuth 1 1 1
Melting Point 258º F. 283º F. 311º F.
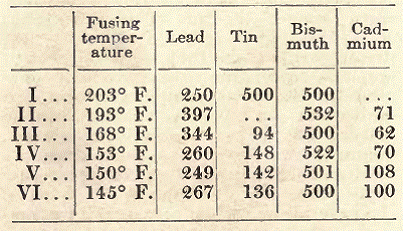
Fusible Alloys for Electric Installations. These alloys are employed in electric installations as current interrupters. Serving as conductors on a short length of circuit, they melt as soon as the current becomes too strong. Following is the composition of some of these alloys.
[65]
ALLOYS
These alloys are prepared by melting the lead in a stearine bath and adding successively, and during the cooling, first, the cadmium; second, the bismuth; third, the tin. It is absolutely necessary to proceed in this manner, since these metals fuse at temperatures ranging from 850º F. (for lead), to 551º F. (for tin).
Fusible Safety Alloys for Steam Boilers.
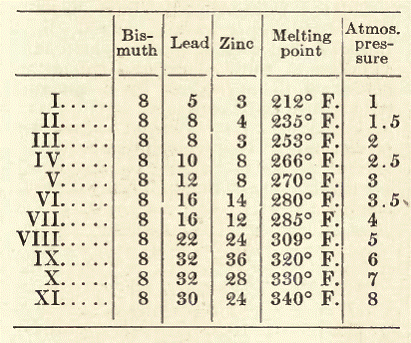
Lipowitz Metal. This amalgam is prepared as follows: Melt in a dish, cadmium, 3 parts, by weight; tin, 4 parts; bismuth, 15 parts; and lead, 8 parts, adding to the alloy, while still in fusion, 2 parts of quicksilver previously heated to about 212º F. The amalgamation proceeds easily and smoothly. The liquid mass in the dish, which should be taken from the fire immediately upon the introduction of the mercury, is stirred until the contents solidify. While Lipowitz alloy softens already at 140º F. and fuses perfectly at 158, the amalgam has a still lower fusing point, which lies around 143|º F.
This amalgam is excellently adapted for the production of impressions of various objects of nature, direct impressions of leaves, and other delicate parts of plants having been made with its aid which, in point of sharpness, are equal to the best plaster casts and have a very pleasing appearance. The amalgam has a silver- white color and a fine gloss. It is perfectly constant to atmospheric influences. This amalgam has also been used with good success for the making of small statuettes and busts, which are hollow and can be readily gilt or bronzed by electro-deposition. The production of small statues is successfully carried out by making a hollow gypsum mold of the articles to be cast and heating the mold evenly to about 140º F. A corresponding quantity of the molten amalgam is then poured in and the mold moved rapidly to and fro, so that the alloy is thrown against the sides all over. The shaking should be continued until it is certain that the amalgam has solidified. When the mold has cooled off it is taken apart and the seams removed by means of a sharp knife. If the operation is carried on correctly, a chasing of the cast mass becomes unnecessary, since the alloy fills out the finest depressions of the mold with the greatest sharpness.
Amalgam for Plaster. Tin, 1 part; bismuth, 1 part; mercury, 1 part. Melt the bismuth and the tin together, and when the two metals are in fusion add the mercury while stirring. For use, rub up the amalgam with a little white of egg and brush like a varnish on the plaster articles.
Plastic Metal Composition.
I. Copper oxide is reduced by means of hydrogen or copper sulphate by boiling a solution of the same in water with some zinc filings in order to obtain entirely pure copper. Of the copper powder obtained in this manner, 20, 30, or 36 parts, by weight, according to the degree of hardness desired for the composition (the greater the quantity of copper used the harder will the composition become), are thoroughly moistened in a cast-iron or porcelain mortar with sulphuric acid of 1.85 specific gravity; 70 parts, by weight, of mercury are then added to this paste, the whole being constantly stirred. When all the copper has been thoroughly amalgamated with the mercury, the sulphuric acid is washed out again with boiling water, and in 12 hours after it has become cold the composition will be so hard that it can be polished. It is impervious to the action of dilute acids, alcohol, ether, and boiling water. It contains the same specific gravity, alike in the soft or the hard condition. When used as a cement, it can at any time be rendered soft and plastic in the following manner: If applied while hot and plastic to the deoxidized surfaces of two pieces of metal, these latter will unite so firmly that in about 10 or 12 hours the metal may be subjected to any mechanical process. The properties of this composition render it very useful for various purposes, and it forms a most effective cement for fine metal articles which cannot be soldered in fire.
II. Bismuth, 5.5 parts; lead, 3; tin, 1.5.
III. Alloy d'Homburg. Bismuth,
[66]
ALLOYS
3 parts; lead, 3; tin, 3. This alloy is fusible at 251º F., and is of a silvery white. It is employed for reproductions of medals.
IV. Alloy Valentine Rose. Bismuth, 4 to 6 parts; lead, 2 parts; tin, 2 to 3 parts. This alloy fuses at 212 to 250º F.
V. Alloy Rose pere. Bismuth, 2 parts; lead, 2; tin, 2. This alloy fuses at 199º F.
The remainder are plastic alloys for reproducing cuts, medals, coins, etc.:
VI. Bismuth, 4 parts; lead, 2 parts; tin, 1 part.
VII. Bismuth, 3 parts; lead, 3 parts; tin, 2 parts.
VIII. Bismuth, 4 parts; lead, 2 parts; tin, 2 parts.
IX. Bismuth, 5 parts; lead, 2 parts; tin, 3 parts.
X. Bismuth, 2 parts; lead, 2 parts; tin, 2 parts.
Quick-Water. That the amalgam may easily take hold of bronze objects and remain there, it is customary to cover the perfectly cleansed and shining article with a thin coat of mercury, which is usually accomplished by dipping it into a so-called quick-water bath.
In the form of minute globules the mercury immediately separates itself from the solution and clings to the bronze object, which thereupon presents the appearance of being plated with silver. After it has been well rinsed in clean water, the amalgam may be evenly and without difficulty applied with the scratch brush.
This quick-water (in reality a solution of mercurous nitrate), is made in the simplest manner by taking 10 parts of mercury and pouring over it 11 parts of nitric acid of a specific gravity equal to 1.33; now let it stand until every part of the mercury is dissolved; then, while stirring vigorously, add 540 parts of water. This solution must be kept in closed flasks or bottles to prevent impurities, such as dust, etc., from falling into it.
The preparatory work on the object to be gilded consists mainly in cleansing it from every trace of oxidation. First, it must be well annealed by placing it in a bed of glowing coal, care being exercised that the heating be uniform. When cooled, this piece is plunged into a highly diluted sulphuric-acid bath in order to dissolve in a measure the oxide. Next it is dipped in a 36 nitric-acid bath, of a specific gravity equal to 1.33, and brushed off with a long brush; it is now dipped into nitric acid into which a little lampblack and table salt have been thrown. It is now ready for washing in clean water and drying in unsoiled sawdust. It is of the greatest importance that the surface to be gilded should appear of a pale yellow tint all over. If it be too smooth the gold will not take hold easily, and if it be too dull it will require too much gold to cover it.
GOLD ALLOYS:
Colored Gold Alloys. The alloys of gold with copper have a reddish tinge; those of gold with silver are whiter, and an alloy of gold, silver, and copper together is distinguished by a greenish tone. Manufacturers of gold ware make use of these different colors, one piece being frequently composed of several pieces of varying color. Below are given some of these alloys, with their colors:
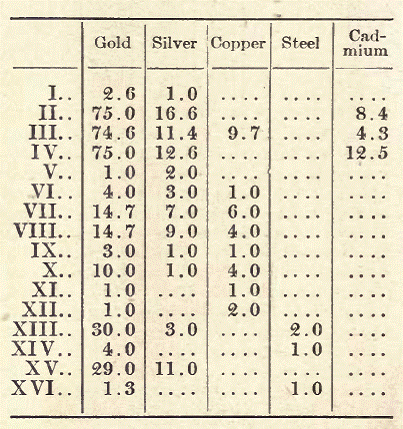
Nos. I, II, III, and IV are green gold; No. Vis pale yellow; Nos. VI, VII, and VIII bright yellow; Nos. IX and X pale red; Nos. XI and XII bright red; Nos. XIII, XIV, and XV gray; while No. XVI exhibits a bluish tint. The finished gold ware, before being put upon the market, is subjected to a special treatment, consisting either in the simple pickling or in the so-called coloring, which operation is conducted especially with alloys of low degree of fineness, the object being to give the layers a superficial layer of pure gold.
The presence of silver considerably modifies the color of gold, and the jeweler makes use of this property to obtain alloys of various shades. The following proportions are to be observed, viz.:
[67]
ALLOYS
Gold Silver Copper
Color of Gold per per per
1,000 1,000 1,000
I. Green 750 250 ...
II. Dead leaves 700 300 ...
III. Sea green 6OO 400 ...
IV. Pink 750 200 50
V. English yellow 750 125 125
VI. English white 750 150 100
VII. Whiter 750 170 80
VIII. Less white 750 190 60
IX. Red 750 ... 250
Other colored gold alloys are the following:
X. Blue. Fine gold, 75; iron, 25.
XI. Dark Gray. Fine gold, 94; iron, 6.
XII. Pale Gray. Fine gold, 191; iron, 9.
XIII. Cassel Yellow. Fine gold, 75; fine silver, 12A; rose copper, 12 1/2.
The above figures are understood to be by weight.
The gold solders, known in France under the names of soudures au quart
(13 1/2 carat), au tiers (12 carat), and au deux (9 carat), are composed of 3, 2, or 1 part of gold respectively, with 1 part of an alloy consisting of two-thirds silver and one-third copper. Gold also forms with aluminum a series of alloys of greatly varying coloration, the most curious of them, composed of 22 parts of aluminum for 88 parts of gold, possessing a pretty purple shade. But all these alloys, of a highly crystalline base, are very brittle and cannot be worked, for which reason their handsome colorings have not yet been capable of being utilized.
Enameling Alloys.
I. Transparent. This alloy should possess the property of transmitting rays of light so as to give the highest possible effect to the enamel. The alloy of gold for transparent green should be pale; a red or copper alloy does not do for green enamel, the copper has a tendency to darken the color and thus take away a part of its brilliancy. The following alloy for transparent green possesses about the nearest print, in color, to the enamel which should represent, as near as possible, the color and brilliancy of the emerald that can be arrived at:
ozs. dwts. grs.
Fine gold 0 18 8
Fine silver 0 1 6
Fine copper 0 0 10
No borax must be used in the melting of this alloy, it being of a more fusible nature than the ordinary alloy, and will not take so high a heat in enameling.
II. Red Enamel. The enamel which forms this color being of a higher fusing point, if proper care be not taken, the gold will melt first, and the work become ruined. In the preparation of red enamel, the coloring matter is usually an oxide of gold, and this so raises the temperature at which it melts that, in order to prevent any mishap, the gold to be enameled on should be what is called a 22-carat red, that is, it should contain a preponderance of copper in the alloying mixture so as to raise the fusing point of the gold. The formula is:
ozs. dwts. grs.
Fine gold 0 18 8
Fine silver 0 0 10
Fine copper 0 1 6
Gold-leaf Alloys. All gold made into leaf is more or less alloyed. The gold used by the goldbeater is alloyed according to the variety of color required. Fine gold is commonly supposed to be incapable of being reduced to thin leaves. This, however, is not the case, although its use for ordinary purposes is undesirable on account of its greater cost. It also adheres by contact of one leaf with another, thus causing spoiled material and wasted labor; but for work exposed to the weather it is much preferable, as it is more durable and does not tarnish or change color.
The following is a list of the principal classes of leaf recognized and ordinarily prepared by beaters with the proportion of alloy they contain:
Gold Silver Copper
grs. grs. grs.
I. Red gold 456-460 ... 0-24
II. Pale red 464 ... 16
III. Extra deep. 456 12 12
IV. Deep 444 24 12
V. Citron 440 30 10
VI. Yellow 408 72 ....
VII. Pale yellow 384 96 ....
VIII. Lemon 360 120 ....
IX. Green or pale 312 168 ....
X. White 240 240 ....
Gold-Plate Alloys. Gold, 92 parts; copper, 8 parts.
II. Gold, 84 parts; copper, 16 parts. III. Gold, 75 parts; copper, 25 parts.
IMITATION GOLD.
I. One hundred parts, by weight, of copper of the purest quality; 14 of zinc or tin; 6 of magnesia; 3/6 of sal ammoniac, limestone, and cream of tartar. The copper is first melted, then the magnesia, sal ammoniac, limestone, and cream of tartar in powder are added separately and gradually. The whole mass is kept stirred for a half hour, the zinc or tin being dropped in piece by piece, the stir-
[68]
ALLOYS
ring being kept up till they melt. Finally the crucible is covered and the mass is kept in fusion 35 minutes and, the same being removed, the metal is poured into molds, and is then ready for use. The alloy thus made is said to be fine-grained, malleable, takes a high polish, and does not easily oxidize.
II. An invention, patented in Germany, covers a metallic alloy, to take the place of gold, which, even if exposed for some time to the action of ammoniacal and acid vapors, does not oxidize or lose its gold color. It can be rolled and worked Tike gold and has the appearance of genuine gold without containing the slightest admixture of that metal. The alloy consists of copper and antimony in the approximate ratio of 100 to 6, and is produced by adding to molten copper, as soon as it has reached a certain degree of heat, the said percentage of antimony.
When the antimony has likewise melted and entered into intimate union with the copper, some charcoal ashes, magnesium, and lime spar are added to the mass when the latter is still in the crucible.
III. Aluminum Gold. This alloy, called Nuremberg gold, is used for making cheap gold ware, and is excellent for this purpose, as its color is exactly that of pure gold, and does not change in the air.
Articles made of Nuremberg gold need no gilding, and retain their color under the hardest usage; even the fracture of this alloy shows the pure gold color. The composition is usually 90 parts of copper, 2.5 of gold, and 7.5 of aluminum.
IV. Imitation gold, capable of being worked and drawn into wire, consists of 950 parts copper, 45 aluminum, and 2 to 5 of silver.
V. Chrysochalk is similar in composition to Mannheim gold:
I II
Copper 90.5 58.68
Zinc 7.9 40.22
Lead 1.6 1.90
In color it resembles gold, but quickly loses its beauty if exposed to the air, on account of the oxidation of the copper. It can, however, be kept bright for a long time by a coating of colorless varnish, which excludes the air and prevents oxidation. Chrysochalk is used for most of the ordinary imitations of gold. Cheap watch chains and jewelry are manufactured from it, and it is widely used by the manufacturers of imitation bronze ornaments.
Mannheim Gold or Similor. Mannheim gold is composed of copper, zinc, and tin, in proportions about as follows:
I II
Copper 83.7 89.8
Zinc 9.3 9.9
Tin 7.0 0.6
It has a fine yellow color, and was formerly much used in making buttons and pressed articles resembling gold. Later alloys, however, surpass it in color, and it has fallen somewhat into disuse.
One variety of Mannheim gold, so called, contains 1.40 parts of brass
(composition 3 Cu 2 1 Zn) to 10 of copper and 0.1 of zinc.
Mosaic Gold. This is an alloy composed with slight deviations of 100 parts of copper and 50 to 55 of zinc. It has a beautiful color, closely resembling that of gold, and is distinguished by a very fine grain, which makes it especially suitable for the manufacture of castings which are afterwards to be gilded. The best method of obtaining a thoroughly homogeneous mixture of the two metals is first to put into the crucible one-half of the zinc to be used, place the cover upon it, and fuse the mixture under a cover of borax at as low a temperature as possible. Have ready the other half of the zinc, cut into small pieces and heated almost to melting, and when the contents of the crucible are liquid throw it in, a small portion at a time, stirring constantly to effect as intimate a mixture of the metals as possible.
Oreiide or Oroide (French Gold). The so-called French gold, when polished, so closely resembles genuine gold in color that it can scarcely be distinguished from it. Besides its beautiful color, it has the valuable properties of being very ductile and tenacious, so that it can easily be stamped into any desired shape; it also takes a high polish. It is frequently used for the manufacture of spoons, forks, etc., but is unsuitable for this purpose on account of the large amount of copper contained in it, rendering it injurious to health. The directions for preparing this alloy vary greatly. The products of some Paris factories show the following composition:
I II III
Copper 90 80.5 86.21
Zinc 10 14.5 31.52
Tin 0.48
Iron 0.24
A special receipt for oreïde is the following:
IV. Melt 100 parts of copper and add, with constant stirring, 6 parts of magnesia, 3.6 of sal ammoniac, 1.8 of lime, and 9 of crude tartar. Stir again
[69]
ALLOYS
thoroughly, and add 17 parts of granulated zinc, and after mixing it with the copper by vigorous stirring keep the alloy liquid for one hour. Then carefully remove the scum and pour off the alloy.
Pinchbeck. This was first manufactured in England. Its dark gold color is the best imitation of gold alloyed with copper. Being very ductile, it can easily be rolled out into thin plates, which can be given any desired shape by stamping. It does not readily oxidize, and thus fulfills all the requirements for making cheap jewelry, which is its principal use.
Copper 88.8 93.6
Zinc 11.2 6.4
Or
Copper 2.1 1.28
Zinc ... 0.7
Brass 1.0 0.7
Palladium Gold. Alloys of gold, copper, silver, and palladium have a brownish-red color and are nearly as hard as iron. They are sometimes (although rarely) used for the bearings for the axles of the wheels of fine watches, as they invite little friction and do not rust in the air. The composition used in the Swiss and English watch factories consists usually of gold 18 parts, copper 13 parts, silver 11, and palladium 6.
Talmi Gold. The name of talmi gold was first applied to articles of jewelry, chains, earrings, bracelets, etc., brought from Paris, and distinguished by beautiful workmanship, a low price, and great durability. Later, when this alloy had acquired a considerable reputation, articles were introduced under the same name, but which were really made of other metals, and which retained their beautiful gold color only as long as they were not used. The fine varieties of talmi gold are manufactured from brass, copper, or tombac, covered with a thin plate of gold, combined with the base by rolling, under strong pressure. The plates are then rolled out by passing through rollers, and the coating not only acquires considerable density, but adheres so closely to the base that the metal will keep its beautiful appearance for years. Of late, many articles of talmi gold have been introduced hose gold coating is produced by electroplating, and is in many cases so thin that hard rubbing will bring through the color of the base. Such articles, of course, are not durable. In genuine talmi gold, the coating, even though it may be thin, adheres very closely to the base, for the reason that the two metals are actually welded by the rolling, and also because alloyed gold is always used, which is much harder than pure gold. The pure gold of electroplating is very soft. The composition of some varieties of talmi gold are here given. It will be seen that the content of gold varies greatly, and the durability of the alloy will, of course, correspond to this. The alloys I, II, III are genuine Paris talmi gold; IV, V, and VI are electroplated imitations; and VII is an alloy of a wrong composition, to which the gold does not adhere firmly:
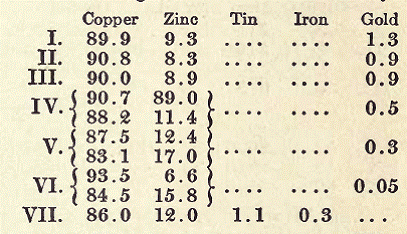
Japanese Alloys. In Japan some specialties in metallic alloys are in use of which the composition is as follows:
Shadke consists of copper with from 1 to 10 per cent of gold. Articles made from this alloy are laid in a pickle of blue vitriol, alum, and verdigris, until they acquire a bluish-black color.
Gui-shi-bu-ichi is an alloy of copper containing 30 to 50 per cent of silver. It possesses a peculiar gray shade.
Mokume consists of several compositions. Thus, about 30 gold foils (gen
uine) are welded together with shadke, copper, silver, and gui-shi-bu-ichi and pierced. The pierced holes are, after firmly hammering together the plates, filled up with the above-named pickle.
The finest Japanese brass consists of 10 parts copper and 8 parts zinc, and is called siachu. The bell metal kara kane is composed of copper 10 parts, tin 10 parts, iron 0.5 part, and zinc 1.5 parts. The copper is first fused, then tne remaining metals are added in rotation.
GERMAN SILVER OR ARGENTAN.
The composition of this alloy varies considerably, but from the adjoined figures an average may be found, which will represent, approximately, the normal composition:
Copper 50 to 66 parts
Zinc 19 to 31 parts
Nickel 13 to 18 parts
The properties of the different kinds, such as their color, ductility, fusibility,
[70]
ALLOYS
etc., vary with the proportions of the single metals. For making spoons, forks, cups, candlesticks, etc., the most suitable proportions are 50 parts of copper, 25 of zinc, and 25 of nickel. This metal has a beautiful blue-white color, and does not tarnish easily.
German silver is sometimes so brittle that a spoon, if allowed to fall upon the floor, will break; this, of course, indicates faulty composition. But the following table will snow how the character of the alloy changes with the varying percentage of the metals composing it:
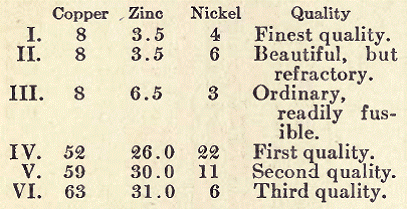
The following analyses give further particulars in regard to different kinds of German silver:
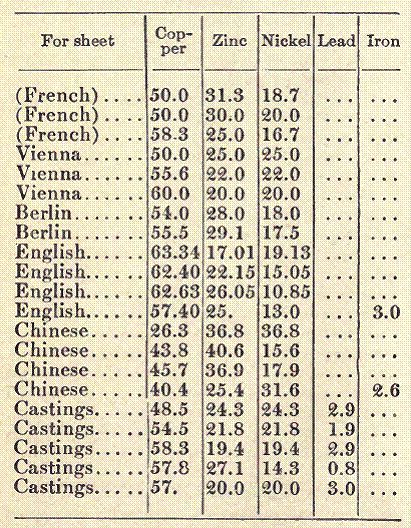
In some kinds of German silver are found varying quantities of iron, manganese, tin, and very frequently lead, added for the purpose of changing the properties of the alloy or cheapening the cost of production. But all these metals have a detrimental rather than a beneficial effect upon the general character of the alloy, and especially lessen its power of resistance to the action of dilute acids, one of its most valuable properties. Lead makes it more fusible; tin acts somewhat as in bronze, making it denser and more resonant, and enabling it to take a higher polish. With iron or manganese the alloy is whiter, but it becomes at the same time more refractory and its tendency toward brittleness is increased.
SUBSTITUTES FOR GERMAN SILVER.
There are many formulas for alloys which claim to be substitutes for German silver; but no one of them has yet become an article of general commerce. It will be sufficient to note these materials briefly, giving the composition of the most important.
Nickel Bronze. This is prepared by fusing together very highly purified nickel (99.5 per cent) with copper, tin, arid zinc. A bronze is produced containing 20 per cent of nickel, light-colored and very hard.
Bismuth Bronze.
I II II IV
Copper 25.0 45.0 69.0 47.0
Nickel 24.0 32.5 10.0 30.9
Antimony 50.0
Bismuth 1.0 1.0 1.0 0.1
Tin 16.0 15.0 1.0
Zinc 21.5 20.0 21.0
Aluminum 1.0
I is hard and very lustrous, suitable for lamp reflectors and axle bearings; II is hard, resonant, and not affected by sea water, for parts of ships, pipes, telegraph wires, and piano strings; III and
IV are for cups, spoons, etc.
Manganese Argentan.
Copper 52 to 50 parts
Nickel 17 to 15 "
Zinc 5 to 10 "
Manganese 1 to 5 "
Copper, with 15 per cent phosphorus 3 to 5 "
Readily cast for objects of art.
Aphtite.
Iron 66 parts
Nickel 23 "
Tungsten 4 "
Copper 5 "
Arguzoid.
Copper 55.78 parts
Zinc 23.198 "
Nickel 13.406 "
Tin 4.035 "
Lead 3.544 "
Silver white, almost ductile, suited for artistic purposes.
[71]
ALLOYS
Ferro - Argentan.
Copper 70.0 parts
Nickel 20.0 "
Zinc 5.5 "
Cadmium 4.5 "
Resembles silver; worked like German silver.
Silver Bronze. Manganese, 18 per cent; aluminum, 1.2 per cent; silicium, 5 per cent; zinc, 13 per cent; copper, 67.5 per cent. The electric resistance of silver bronze is greater than that of German silver, hence it ought to be highly suitable for rheostats.
Instrument Alloys. The following are suitable for physical and optical instruments, metallic mirrors, telescopes, etc.:
I. Copper, 62 parts; tin, 33 parts; lead, 5 parts.
II. Copper, 80; antimony, 11; lead, 9.
III. Copper, 10; tin, 10; antimony, 10; lead, 40.
IV. Copper, 30; tin, 50; silver, 2; arsenic, 1.
V. Copper, 66; tin, 33.
VI. Copper, 64; tin, 26.
VII. Steel, 90; nickel, 10.
VIII. Platinum, 60; copper, 40.
IX. Platinum, 45; steel, 55.
X. Platinum, 55; iron, 45.
XI. Platinum, 15; steel, 85.
XII. Platinum, 20; copper, 79; arsenic, 1.
XIII. Platinum, 62; iron, 28; gold, 10.
XIV. Gold, 48; zinc, 52.
XV. Steel, 50; rhodium, 50.
XVI. Platinum, 12; iridium, 88.
XVII. Copper, 89.5; tin, 8.5; zinc, 2.
LEAD ALLOYS.
The following alloys, principally lead, are used for various purposes:
Bibra Alloy. This contains 8 parts of bismuth, 9 of tin, and 38 to 40 of lead.
Metallic Coffins. Tin, 40 parts; lead, 45 parts; copper, 15 parts.
Plates for Engraving.
I. Lead, 84 parts; antimony, 16 parts.
II. Lead, 86 parts; antimony, 14 parts.
III. Lead, 87 parts; antimony, 12 parts; copper, 1 part.
IV. Lead, 81 parts; antimony, 14 parts; tin, 5 parts.
V. Lead, 73 parts; antimony, 17 parts; zinc, 10 parts.
VI. Tin, 53 parts; lead, 43 parts; antimony, 4 parts.
Hard lead is made of lead, 84 parts; antimony, 16 parts.
Sheet Metal Alloy.
Tin 35 parts
Lead 250 parts
Copper 2.5 parts
Zinc 0.5 part
This alloy has a fine white color, and can be readily rolled into thin sheets. For that reason it is well adapted for lining tea chests and for the production of tobacco and chocolate wrappers. The copper and zinc are used in the form of fine shavings. The alloy should be immediately cast into thin plates, which can then be passed through rolls.
MAGNETIC ALLOYS.
Alloys which can be magnetized most strongly are composed of copper, manganese, and aluminum, the quantities of manganese and aluminum being proportional to their atomic weights (55.0 to 27.1, or about 2 to 1). The maximum magnetization increases rapidly with increase of manganese, but alloys containing much manganese are exceedingly brittle and cannot be wrought. The highest practicable proportion of manganese at present is 24 per cent.
These magnetic alloys were studied by Hensler, Haupt, and Starck, and Gumlich has recently examined them at the Physikalisch-technische Reichsanstalt, with very remarkable and interesting results.
The two alloys examined were composed as follows:
Alloy I. Copper, 61.5 per cent; manganese, 23.5 per cent; aluminum, 15 per cent; lead, 0.1 per cent, with traces of iron and silicon.
Alloy II. Copper, 67.7 per cent; manganese, 20.5 per cent; aluminum,
10.7 per cent; lead, 1.2 per cent, with traces of iron and silicon.
Alloy II could be worked without difficulty, but alloy I was so brittle that it broke under the hammer. A bar 7 inches long and 1/4 inch thick was obtained by grinding. This broke in two during the measurements, but, fortunately, without invalidating them. Such a material is evidently unsuited to practical uses.
The behavior of magnetic alloys at high temperatures is very peculiar. Alloy I is indifferent to temperature changes, which scarcely affect its magnetic properties, but the behavior of alloy II is very different. Prolonged heating to 230º F. produces a great increase in its capability of magnetization, which, after 544 hours' heating, rises from 1.9 to 3.2 kilo-
[72]
ALLOYS
gauss, approaching the strength of alloy I. But when alloy II is heated to 329º F., its capability of magnetization fails again and the material suffers permanent injury, which can be partly, but not wholly, cured by prolonged heating.
Another singular phenomenon was exhibited by both of these alloys. When a bar of iron is magnetized by an electric current, it acquires its full magnetic strength almost instantaneously on the closure of the circuit. The magnetic alloys, on the contrary, do not attain their full magnetization for several minutes. In some of the experiments a gradual increase was observed even after the current had been flowing five minutes. In magnetic strength alloy I proved far superior to alloy II, which contained smaller proportions of manganese and aluminum. Alloy I showed magnetic strengths up to 4.5 kilogauss, while the highest magnetization obtained with alloy II was only 1.9 kilogauss. But even alloy II may be called strongly magnetic, for its maximum magnetization is about one-tenth that of good wrought iron (18 to 20 kilogauss), or one-sixth that of cast iron (10 to 12 kilogauss). Alloy I is nearly equal in magnetic properties to nickel, which can be magnetized up to about 5 kilogauss.
MANGANESE ALLOYS:
Manganese bronze is a bronze deprived of its oxide by an admixture of manganese. The manganese is used as copper manganese containing 10 to 30 per cent manganese and added to the bronze to the amount of 0.5 to 2 per cent.
Manganese Copper. The alloys of copper with manganese have a beautiful silvery color, considerable ductility, great hardness and tenacity, and are more readily fusible than ordinary bronze. A special characteristic is that they exactly fill out the molds, without the formation of blowholes, and present no difficulties in casting.
Cupromanganese is suitable for many purposes for which nothing else but bronze can advantageously be used, and the cost of its production is no greater than that of genuine bronze. In preparing the alloy, the copper is used in the form of fine grains, obtained by pouring melted copper into cold water. These copper grains are mixed with the dry oxide of manganese, and the mixture put into a crucible holding about 66 pounds.
Enough space must be left in the crucible to allow a thick cover of charcoal, as the manganese oxidizes easily. The crucible is placed in a well-drawing wind furnace and subjected to a strong white heat. The oxide of manganese is completely reduced to manganese, which at once combines with the copper to form an alloy. In order to prevent, as far as possible, the access of air to the fusing mass, it is advisable to cover the crucible with a lid which has an aperture in the center for the escape of the carbonic oxide formed during the reduction.
When the reduction is complete and the metals fused, the lid is removed and the contents of the crucible stirred with an iron rod, in order to make the alloy as homogeneous as possible. By repeated remelting of the cupromanganese a considerable quantity of the manganese is reconverted into oxide; it is, therefore, advisable to make the casts directly from the crucible. When poured out, the alloy rapidly solidifies, and resembles in appearance good German silver. Another reason for avoiding remelting is that the crucible is strongly attacked by the cupromanganese, and can be used but a few times.
The best kinds of cupromanganese contain between 10 and 30 per cent of manganese. They have a beautiful white color, are hard, tougher than copper, and can be worked under the hammer or with rolls. Some varieties of cupromanganese which are especially valuable for technical purposes are given below:
I II III IV
Copper 75 60 65 60
Manganese 25 25 20 20
Zinc 15 5
Tin 10
Nickel 10 10
Manganin. This is an alloy of copper, nickel, and manganese for electric resistances.
MIRROR ALLOYS:
Amalgams for Mirrors.
I. Tin, 70 parts; mercury, 30 parts.
II. For curved mirrors. Tin, 1 part; lead, 1 part; bismuth, 1 part; mercury, 9 parts.
III. For glass balls. Tin, 80 parts; mercury, 20 parts.
IV. Metallic cement. Copper, 30 parts; mercury, 70 parts.
V. Mirror metal. Copper, 100 parts; tin, 50 parts; Chinese copper, 8 parts; lead, 1 part; antimony, I part.
Reflector Metals.
I. (Cooper's.) Copper, 35 parts; platinum, 6; zinc, 2; tin, 16.5; arsenic, 1. On account of the hardness of this alloy, it takes a very high polish; it is impervious to the effects of the weather, and is therefore remark-
[73]
ALLOYS
ably well adapted to the manufacture of mirrors for fine optical instruments.
II. (Duppler's.) Zinc, 20 parts; silver, 80 parts.
III. Copper, 66.22 parts; tin, 33.11 parts; arsenic, 0.67 part.
IV. Copper, 64 parts; tin, 32 parts; arsenic, 4 parts.
V. Copper, 82.18 parts; lead, 9.22 parts; antimony, 8.60 parts.
VI. (Little's.) Copper, 69.01 parts; tin, 30.82 parts; zinc, 2.44 parts; arsenic, 1.83 parts.
Speculum Metal. Alloys consisting of 2 parts of copper and 1 of tin can be very brilliantly polished, and will serve for mirrors. Good speculum metal should have a very fine-grained fracture, should be white and very hard, the highest degree of polish depending upon these qualities. A composition to meet these requirements must contain at least 35 to 36 per cent of copper. Attempts have frequently been made to increase the hardness of speculum metal by additions of nickel, antimony, and arsenic. With the exception of nickel, these substances have the effect of causing the metal to lose its high luster easily, any considerable quantity of arsenic in particular having this effect.
The real speculum metal seems to be a combination of the formula Cu4Sn,
composed of copper 68.21 per cent, tin 31.7. An alloy of this nature is sometimes separated from ordnance bronze by incorrect treatment, causing the socalled tin spots; but this has not the pure white color which distinguishes the speculum metal containing 31.5 per cent of tin.
By increasing the percentage of copper the color gradually shades into yellow; with a larger amount of tin into blue. It is dangerous to increase the tin too much, as this changes the other properties of the alloy, and it becomes too brittle to be worked. Below is a table showing different compositions of speculum metal. The standard alloy is undoubtedly the best.
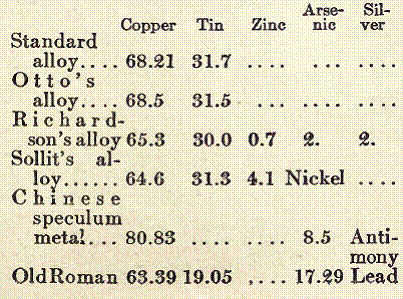
PALLADIUM ALLOYS.
I. An alloy of palladium 24 parts, gold 80, is white, hard as steel, unchangeable in the air, and can, like the other alloys of palladium, be used for dental purposes.
II. Palladium 6 parts, gold 18, silver 11, and copper 13, gives a reddish-brown, hard, and very fine-grained alloy, suitable for the bearings of pivots in clock works.
The alloys of most of the other platinum metals, so called, are little used on account of their rarity and costliness. Iridium and rhodium give great hardness to steel, but the commercial rhodium and iridium steel, so called, frequently contains not a trace of either. The alloy of iridium with osmium has great hardness and resistance and is recommended for pivots, fine instruments, and points of ship compasses.
Palladium Silver. This alloy, composed of 9 parts of palladium and 1 of silver, is used almost exclusively for dental purposes, and is well suited to the manufacture of artificial teeth, as it does not oxidize. An alloy even more frequently used than this consists of platinum 10 parts, palladium 8, and gold 6.
Palladium Bearing Metal. This alloy is extremely hard, and is used instead of jewel bearings in watches. It is composed of palladium 24 parts, gold 72, silver 44, copper 92.
PLATINUM ALLOYS.
Platinum has usually been alloyed with silver in goldsmith's work, 2 parts silver to 1 of platinum being taken to form the favorite "platinum silver." The object has been to produce an alloy having a white appearance, which can be polished, and at the same time has a low melting point. In addition to this Elatinum alloy the following are well known:
I. A mixture of 7 parts platinum with 3 parts iridium. This gives to platinum the hardness of steel, which can be still further increased by taking 4 parts of iridium.
II. An alloy of 9 parts platinum and 1 part iridium is used by the French in the manufacture of measuring instruments of great resisting power.
Compounds of copper, nickel, cadmium, and tungsten are also used in the
construction of parts of watches; the latter acquire considerable hardness without becoming magnetic or rusting like steel.
III. For this purpose a compound of
[74]
ALLOYS
62.75 parts platinum, 18 parts copper, 1.25 parts cadmium, and 18 parts nickel is much recommended.
IV. Very ductile platinum-copper alloys have also been made, e. g., the so-called Cooper gold, consisting of 3 parts platinum and 13 parts copper, which is almost equal to 18-carat gold in regard to color, finish, and ductility. If 4 per cent of platinum is taken, these latter alloys acquire a rose-red color, while a golden-yellow color can be produced by further adding from 1 to 2 per cent (in all 5 to 6 per cent) of platinum. The last-named alloy is extensively used for ornaments, likewise alloy V.
V. Ten parts platinum, 60 parts nickel, and 220 parts brass, or 2 parts platinum, 1 part nickel and silver respectively, 2 parts brass, and 5 parts copper; this also gives a golden-yellow color.
VI. For table utensils a favorite alloy is composed of 1 part platinum, 100 parts nickel, and 10 parts tin. Articles made of the latter alloy are impervious to atmospheric action and keep their polish for a long time. Pure white platinum alloys have for some time been used in dental work, and they have also proved serviceable for jewelry.
VII. A mixture of 30 parts platinum, 10 parts gold, and 3 parts silver, or 7 parts platinum, 2 parts gold, and 3 parts silver.
VIII. For enameled articles: Platinum, 35 parts; silver, 65 parts. First fuse the silver, then add the platinum in the spongy form. A good solder for this is platinum 80 parts, copper 20 parts.
IX. For pens: Platinum, 4 parts; silver, 3 parts; copper, 1 part.
Platinum Gold. Small quantities of platinum change the characteristics of gold in many respects. With a small percentage the color is noticeably lighter than that of pure gold, and the alloys are extremely elastic; alloys containing more than 20 per cent of platinum, however, almost entirely lose their elasticity. The melting point of the platinum-gold alloy is high, and alloys containing 70 per cent of platinum can be fused only in the flame of oxyhydrogen gas, like platinum itself. Alloys with a smaller percentage of platinum can be prepared in furnaces, but require the strongest white heat. In order to avoid the chance of an imperfect alloy from too low a temperature, it is always safer to fuse them with the oxyhydrogen flame. The alloys of platinum and gold have a somewhat limited application. Those which contain from 5 to 10 per cent of platinum are used for sheet and wire in the manufacture of artificial teeth.
Platinum-Gold Alloys for Dental Purposes.
I II III
Platinum 6 14 10
Gold 2 4 6
Silver 1 6 -
Palladium - - 8
Platinum Silver. An addition of platinum to silver makes it harder, but also more brittle, and changes the white color to gray. An alloy which contains only a very small percentage of platinum is noticeably darker in color than pure silver. Such alloys are prepared under the name of platine au titre, containing between 17 and 35 per cent of platinum. They are almost exclusively used for dental purposes.
Imitation Platinum.
I. Brass, 100 parts; zinc, 65 parts.
II. Brass, 120 parts; zinc, 75 parts.
III. Copper, 5 parts; nickel, 4 parts; zinc, l 1/2 parts; antimony, 1 part; lead, 1 part; iron, 1 part; tin, 1 part.
Cooper's Pen Metal. This alloy is especially well adapted to the manufacture of pens, on account of its great hardness, elasticity, and power of resistance to atmospheric influences, and would certainly have superseded steel if it were possible to produce it more cheaply than is the case. The compositions most frequently used for pen metal are copper 1 part, platinum 4, and silver 3; or, copper 21, platinum 50, and silver 36.
Pens have been manufactured, consisting of several sections, each of a different alloy, suited to the special purpose of the part. Thus, for instance, the sides of the pen are made of the elastic composition just described; the upper part is of an alloy of silver and platinum; and the point is made either of minute cut rubies or of an extremely hard alloy of osmium and iridium, joined to the body of the pen by melting in the flame of the oxyhydrogen blowpipe. The price of such pens, made of expensive materials and at the cost of great labor, is of course exceedingly high, but their excellent qualities repay the extra expense. They are not in the least affected by any kind of ink, are most durable, and can be used constantly for years without showing any signs of wear.
The great hardness and resistance to the atmosphere of Cooper's alloys make them very suitable for manufacturing;
[75]
ALLOYS
mathematical instruments where great precision is required. It can scarcely be calculated how long a chronometer, for instance, whose wheels are constructed of this alloy, will run before showing any irregularities due to wear. In the construction of such instruments, the price of the material is not to be taken into account, since the cost of the labor in their manufacture so far exceeds this.
PEWTER.
This is an alloy of tin and lead only, or of tin with antimony and copper. The first is properly called pewter. Three varieties are known in trade:
I (Plate Pewter). From tin, 79 per cent; antimony, 7 per cent; bismuth and copper, of each 2 per cent; fused together. Used to make plates, teapots, etc. Takes a fine polish.
II (Triple Pewter). From tin, 79 per cent; antimony, 15 per cent; lead, 6 per cent; as the last. Used for minor articles, syringes, toys, etc.
III (Ley Pewter). From tin, 80 per cent; lead, 20 per cent. Used for
measures, inkstands, etc.
According to the report of a French commission, pewter containing more than 18 parts of lead to 82 parts of tin is unsafe for measures for wine and similar liquors, and, indeed, for any other utensils exposed to contact with food or beverages. The legal specific gravity of pewter in France is 7.764; if it be greater, it contains an excess of lead, and is liable to prove poisonous. The proportions of these metals may be approximately determined from the specific gravity; but correctly only by an assay for the purpose.
SILVER ALLOYS:
Aluminum Silver. Aluminum and silver form beautiful white alloys which are considerably harder than pure aluminum, and take a very high polish. They have the advantage over copper alloys of being unchanged by exposure to the air, and of retaining their white color.
The properties of aluminum and silver alloys vary considerably according to the percentage of aluminum.
I. An alloy of 100 parts of aluminum and 5 parts of silver is very similar to pure aluminum, but is harder and takes a finer polish.
II. One hundred and sixty-nine parts of aluminum and 5 of silver make an elastic alloy, recommended for watch springs and dessert knives.
III. An alloy of equal parts of silver and aluminum is as hard as bronze.
IV. Five parts of aluminum and 1 part of silver make an alloy that is easily worked.
V. Also aluminum, 3 parts, and silver, 1 part.
VI. Tiers-Argent. This alloy is prepared chiefly in Paris, and used for the manufacture of various utensils. As indicated by its name (one-third silver), it consists of 33.33 parts of silver and 66.66 parts of aluminum. Its advantages over silver consist in its lower price and greater hardness; it can also be stamped and engraved more easily than the alloys of copper and silver.
VII. This is a hard alloy which has been found very useful for the operating levers of certain machines, such as the spacing lever of a typewriter. The metal now generally used for this purpose by the various typewriter companies is "aluminum silver," or "silver metal." The proportions are given as follows:
Copper 57.00
Nickel 20.00
Zinc 20.00
Aluminum 3.00
This alloy when used on typewriting machines is nickel-plated for the sake of the first appearance, but so far as corrosion is concerned, nickeling is unnecessary. The alloy is stiff and strong and cannot be bent to any extent without breaking, especially if the percentage of aluminum is increased to 3.5 per cent; it casts free from pinholes and blow holes; the liquid metal completely fills the mold, giving sharp, clean castings, true to pattern; its cost is not greater than brass; its color is silver white, and its hardness makes it susceptible to a high polish.
Arsenic. Alloys which contain small quantities of arsenic are very ductile, have a beautiful white color, and were formerly used in England in the manufacture of tableware. They are not, however, suitable for this purpose, on account of the poisonous character of the arsenic. They are composed usually of 49 parts of silver, 49 of copper, and 2 of arsenic.
China Silver. Copper, 65.24 per cent; tin, 19.52 per cent; nickel, 13.00 per cent; silver, 2.05 per cent.
Copper-Silver.
When silver is alloyed with copper only one proportion is
known which will give a uniform casting. The proportion is
72 per cent silver to 28 per cent copper. With more silver
than 72 per cent the center of a cast bar will be