 The
Science Notebook
The
Science NotebookHenley's Book of Formulas, Recipes and Processes
Home Terms of Use Safety Contact Us Experiment Pages Downloads Supplies Useful Links!
Henley's Twentieth Century Book of Formulas, Recipes and Processes - Pages 751-775
[751]
WATERPROOFING
its folds to stick together when it is wrapped up, or in the other extreme to crack, still remains. The weight, of course, is inevitable. An oilskin must be heavy, comparatively, from the very essence of the process by which it is made, but there seems no reason why it should not in time be made much more pliable (an old-time oilskin coat could often stand up on end when empty) and free from the danger of cracking or being compacted into a solid block when it has been stored folded on a shelf.
Probably the best oilskins ever made are those prepared by combining Dr. Stenhouse's process (patented in 1864) with the ordinary method, which consists in the main of painting over the fabric with two or more coats of boiled linseed oil, allowing each coat to dry before the next is applied. This, with a few variations in detail, is the whole method of making oilskins. Dr. Stenhouse's waterproofing method is to impregnate the fabric with a mixture of hard paraffine and boiled oil in proportions varying according to circumstances from 95 per cent of paraffine and 5 of oil to 70 per cent of the former and 30 of the latter. The most usual percentages are 80 and 20. The mixture is made with the aid of heat, and is then cast into blocks for storage. It is applied to the cloth stretched on a hot plate by rubbing the fabric thoroughly all over with a block of the composition, which may be applied on one or both sides as may be wished. The saturation is then made complete, and excess of composition is removed by passing the cloth between hot rollers. When the cloth is quite cold the process is complete. The paraffine and the drying oil combine their waterproofing powers, and the paraffine prevents the oil from exerting any injurious action upon the material. Drying oil, partly on account of the metallic compounds in it, and partly on account of its absorbing oxygen from the atmosphere, has a decided slow weakening effect upon textile fibers.
Dr. Stenhouse points out that the inflammability of oilskins may be much lessened by the use of the ordinary fireproofing salts, such as tungstate of soda, or alum, either before or after the waterproofing process is carried out.
The following are some of the best recommended recipes for making oilskins:
I. Dissolve 1 ounce of yellow soap in 1 1/2 pints of boiling water. Then stir in 1 quart of boiled oil. When cold, add 1/4 pint of gold size.
II. Take fine twilled calico. Soak it in bullock's blood and dry it. Then give it 2 or 3 coats of boiled oil, mixed with a little litharge, or with an ounce of gold size to every pint of the oil.
III. Make ordinary paint ready to be applied thin with a strong solution of soap.
IV. Make 96 pounds of ocher to a thin paste with boiled oil, and then add 16 pounds of ordinary black paint mixed ready for use. Apply the first coat of this with soap, the subsequent coats without soap.
V. Dissolve rosin in hot boiled oil till it begins to thicken.
VI. Mix chalk or pipe clay in the finest powder, and in the purest state obtainable to a thin paste with boiled oil.
VII. Melt together boiled oil, 1 pint; beeswax and rosin, each, 2 ounces.
VIII. Dissolve soft soap in hot water and add solution of protosulphate of iron till no further precipitate is produced. Filter off, wash, and dry, and form the mass into a thin paste with boiled oil.
All these compositions are painted on with an ordinary painter's brush. The fabric should be slightly stretched, both to avoid folds and to facilitate the penetration of the waterproofing mixture. To aid the penetration still further, the mixture should be applied hot. It is of the greatest importance that the fabric should not be damp when the composition is applied to it. It is best to have it warm as well as the composition. If more than one coat is applied, which is practically always the case, three being the usual number, it is essential that the
last coat should be perfectly dry before the next is applied. Neglect of this precaution is the chief cause of stickiness, which frequently results in serious damage to the oilskins when they have to be unfolded. In fact, it is advisable to avoid folding an oilskin when it can be avoided. They should be hung up when not in use, whenever practicable, and be allowed plenty of room. It goes without saying that no attempt should be made to sell or use the oilskin, whether garment or tarpaulin, until the final coat of composition is perfectly dry and set. It is unadvisable to use artificial heat in the drying at any stage in the manufacture.
Waterproofing Paper. Any convenient and appropriate machinery or apparatus may be employed; but the best method for waterproofing paper is as follows: The treatment may be applied
[752]
WATERPROOFING
while the pulp is being formed into paper, or the finished paper may be treated. If the material is to be treated while being formed into paper, then the better method is to begin the treatment when the web of pulpy material leaves the Foudrinier wire or the cylinders, it then being in a damp condition, but with the larger percentage of moisture removed. From this point the treatment of the paper is the same whether it be pulp in a sheet, as above stated, or finished paper.
The treatment consists, first, in saturating the paper with glutinous material, preferably animal glue, and by preference the bath of glutinous material should be hot, to effect the more rapid absorption and more perfect permeation, impregnation, and deposit of the glutinous material within all the microscopic interstices throughout the body of the paper being treated. By preference a suitable tank is provided in which the glutinous material is deposited, and in which it may be kept heated to a constant temperature, the paper being passed through the tank and saturated during its passage. The material being treated should pass in a continuous sheet that is, be fed from a roll and the finished product be wound in a roll after final treatment. This saves time and the patentee finds that the requisite permeation or incorporation of glutinous matter in the fiber will with some papers for instance - lightly sized manila hemp - require but a few seconds. As the paper passes from the glutin tank the surplus of the glutinous matter is removed from the surface by mechanical means, as contradistinguished from simply allowing it to pass off by gravity, and in most instances it is preferred to pass the paper between suitable pressure rolls to remove such surplus. The strength and consistency of the glutinous bath may be varied, depending upon the material being treated and the uses for which such material is designed. It may, however, be stated that, in a majority of cases, a hot solution of about 1 part of animal glue to about 10 parts of water, by weight, gives the best results. After leaving the bath of glutinous material and having the surplus adhering to the surfaces removed, the paper before drying is passed into or through a solution of formaldehyde and water to "set" the glutinous material. The strength of this solution may also be variable, depending, as heretofore stated, upon the paper and uses for which it is designed. In the majority of cases, however, a solution of 1 part of formaldehyde (35 per cent solution) to 5 parts of water, by weight, gives good results, and the best result is attained if this bath is cold instead of hot, though any particular temperature is not essentially necessary. The effect of the formaldehyde solution upon the glutin-saturated paper is to precipitate the glutinous matter and render it insoluble.
As the material comes from the formaldehyde bath, the surplus adhering to the surfaces is removed by mechanical means, pressure rolls being probably most convenient. The paper is then dried in any convenient manner. The best result in drying is attained by the air-blast, i.e, projecting blasts of air against both surfaces of the paper. This drying removes all the watery constituents and leaves the paper in a toughened or greatly strengthened condition, but not in practical condition for commercial uses, as it is brittle, horny, and stiff, and has an objectionable odor and taste on account of the presence of the aldehydes, paraldehydes, formic acid, and other products, the result of oxidation. Hence it needs to be "tempered." Now while the glutinous material is rendered insoluble that is, it is so acted upon by formaldehyde and the chemical action which takes place while the united solutions are giving off their watery constituents that it will not fully dissolve it is, however, in a condition to be acted on by moisture, as it will swell and absorb, or take up permanently by either chemical or mechanical action a percentage of water, and will also become improved in many respects, so that to temper and render the paper soft and pliable and adapt it for most commercial uses it is subjected to moisture, which penetrates the paper, causing a welling in all directions, filling the interstices perfectly and resulting in "hydration" throughout the entire cellular structure. Two actions, mechanical and chemical, appear to take place, the mechanical action being the temporary absorption of water analogous to the absorption of water by a dry sponge, the chemical action being the permanent union of water with the treated paper, analogous to the union of water and tapioca, causing swelling, or like the chemical combination of water with lime or cement. For this purpose it is preferred to pass the paper into a bath of hot water, saturated steam or equivalent heat-and-moisture medium, thus causing the fibers and the non-soluble glutinous material filling the interstices to expand in all directions and forcing
[753]
WATERPROOFING WAX
the glutinous material into all the microscopic pores or openings and into the masses of fiber, causing a commingling or thorough incorporation of the fibers and the glutinous compound. At the same time, as heretofore indicated, a change (hydration) takes place, whereby the hardened mass of fiber, glutinous material, and formaldehyde become tempered and softened and the strength imparted by the previous treatment increased. To heighten the tempering and softening effect, glycerine may, in some instances, be introduced in the tempering bath, and in most cases one two-hundredths in volume of glycerine gives the best results.
The paper may be dried in any convenient manner and is in condition for most commercial uses, it being greatly strengthened, more flexible, more impervious to moisture, acids, grease, or alkalies, and is suitable for the manufacture of binding-twine, carpets, and many novelties, for dry wrappings and lining packing cases, etc., but is liable to have a disagreeable taste and may carry traces of acids, rendering it impracticable for some uses for instance, wrapping butter, meats, cheese, etc., after receiving the alkali treatment. The paper is also valuable as a packing for joints in steam, water, and other pipes or connections. For the purpose, therefore, of rendering the material absolutely free from all traces of acidity and all taste and odors and, in fact, to render it absolutely hygienic, it is passed through a bath of water and a volatile alkali (ammonium hydrate), the proportion by preference in a majority of cases being one-hundredth of ammonium hydrate to ninety-nine one-hundredths of water by volume. A small percentage of wood alcohol may be added. This bath is preferably cool, but a variation in its temperature will not interfere to a serious extent with the results. The effect of this bath followed by drying is to complete the chemical reaction and destroy all taste or odor, removing all traces of acids and rendering the paper hygienic in all respects. The material may be calendered or cut and used for any of the purposes desired. If the material is to be subjected to the volatile alkali bath, it is not necessary to dry it between the tempering and volatile alkali baths.
The paper made in accordance with the foregoing will, it is claimed, be found to be greatly strengthened, some materials being increased in strength from 100 to 700 per cent. It will be nonabsorbent to acids, greases, and alkalies, and substantially waterproof, and owing to its component integrate structure will be practically non-conductive to electricity, adapting it as a superior insulating material. It may with perfect safety be employed for wrapping butter, meats, spices, groceries, and all materials, whether unctuous or otherwise.
The term "hydration" means the subjecting of the material (after treatment with glutinous material and formaldehyde and drying) to moisture, whereby the action described takes place.
A sheet or web of paper can be treated by the process as rapidly as it is manufactured, as the time for exposure to the action of the glutinous material need not be longer than the time required for it to become saturated, this, of course, varying with different thicknesses and densities, and the length of time of exposure may be fixed without checking the speed by making the tank of such length that the requisite time will elapse while the sheet is passing through it and the guides so arranged as to maintain the sheet in position to be acted on by such solution the requisite length of time. Four seconds' exposure to the action of formaldehyde is found sufficient in most cases.
Waterproof Ropes. For making ropes and lines impervious to weather the process of tarring is recommended, which can be done either in the separate strands or after the rope is twisted. An addition of tallow gives greater pliability.
Waterproof Wood.
I. Soak in a mixture of boracic acid, 6 parts; ammonium chloride, 5 parts; sodium borate, 3 parts, and water, 100 parts.
II. Saturate in a solution of zinc chloride.
Wax
Adulteration of Wax. Wax is adulterated with the following among other substances: Rosins, pitch, flowers of sulphur, starch, fecula, stearine, paraffine, tallow, palm oil, calcined bones, yellow ocher, water, and wood sawdust.
Rosins are detected by cold alcohol, which dissolves all rosinous substances and exercises no action on the wax. The rosins having been extracted from the alcoholic solution by the evaporation of the alcohol, the various kinds may be distinguished by the odors disengaged
by burning the mass several times on a plate of heated iron.
All earthy substances may be readily
[754]
WAX
separated frcm wax by means of oil of turpentine, which dissolves the wax, while the earthy matters form a residue.
Oil of turpentine also completely separates wax from starchy substances, which, like earthy matters, do not dissolve, but form a residue. A simpler method consists in heating the wax with boiling water; the gelatinous consistency assumed by the water, and the blue coloration in presence of iodine, indicate that the wax contains starchy substances. Adulteration by means of starch and fecula is quite frequent. These substances are sometimes added to the wax in a proportion of nearly 60 per cent. To separate either, the suspected product is treated hot with very dilute sulphuric acid (2 parts of acid per 100 parts of water). All amylaceous substances, converted into dextrin, remain dissolved in the liquid, while the wax, in cooling, forms a crust on the surface. It is taken off and weighed; the difference between its weight and that of the product analyzed will give the quantity of the amylaceous substances.
Flowers of sulphur are recognized readily from the odor of sulphurous acid during combustion on red-hot iron.
Tallow may be detected by the taste and odor. Pure wax has an aromatic,
agreeable taste, while that mixed with tallow is repulsive both in taste and smell. Pure wax, worked between the fingers, grows soft, preserving a certain cohesion in all parts. It divides into lumps, which adhere to the fingers, if it is mixed with tallow. The adulteration may also be detected by the thick and nauseating fumes produced when it is burned on heated iron.
Stearic acid may be recognized by means of boiling alcohol, which dissolves it in nearly all proportions and causes it to deposit crystals on cooling, while it is without action on the wax. Blue litmus paper, immersed in alcohol solution, reddens on drying in air, and thus serves for detecting the presence of stearic acid.
Ocher is found by treating the wax with boiling water. A lemon-yellow deposit results, which, taken up with chlorhydric acid, yields with ammonia a lemon-yellow precipitate of ferric oxide.
The powder of burnt bones separates and forms a residue, when the wax is heated with oil of turpentine.
Artificial Beeswax. This is obtained by mixing the following substances, in approximately the proportions stated: Paraffine, 45 parts, by weight; white Japan vegetable wax, 30 parts, by weight; rosins, or colophonies, 10 parts, by weight; white pitch, 10 parts, by weight; tallow, 5 parts, by weight; ceresine, colorant, 0.030 parts, by weight; wax perfume, 0.100 parts, by weight. If desired, the paraffine may be replaced with ozokerite, or by a mixture of vaseline and ozokerite, for the purpose of varying the fusing temperature, or rendering it more advantageous for the various applications designed. The following is the method of preparation: Melt on the boiling water bath, shaking constantly, the paraffine, the Japan wax, the rosins, the pitch, and the tallow. When the fusion is complete, add the colorant and the perfume. When these products are perfectly mingled, remove from the fire, allow the mixture to cool, and run it into suitable molds. The wax thus obtained may be employed specially for encaustics for furniture and floors, or for purposes where varnish is employed.
Waxes for Floors, Furniture, etc.
I.
White beeswax 16 parts
Colophony 4 parts
Venice turpentine 1 part
Melt the articles together over a gentle fire, and when completely melted and homogeneous, pour into a sizable earthenware vessel, and stir in, while still warm, 6 parts of the best French turpentine. Cool for 24 hours, by which time the mass has acquired the consistence of soft butter, and is ready for use. Its method of use is very simple. It is smeared, in small quantities, on woolen cloths, and with these is rubbed into the wood.
This is the best preparation, but one in which the beeswax is merely dissolved in the turpentine in such a way as to have the consistence of a not too thin oil color, will answer. The wood is treated with this, taking care that the surface is evenly covered with the mixture, and that it does not sink too deeply in the ornaments, corners, etc., of the woodwork. This is best achieved by taking care to scrape off from the cloths all excess of the wax.
If, in the course of 24 hours, the surface is hard, then with a stiff brush go over it, much after the way of polishing a boot. For the corners and angles smaller brushes are used; when necessary, stiff pencils may be employed. Finally, the whole is polished with plush, or velvet rags, in order not to injure the original polish. Give the article a good coat of linseed oil or a washing with petroleum before beginning work.
II. Articles that are always exposed to the water, floors, doors, especially of oak, should, from time to time, be satu
[755]
WAX
rated with oil or wax. A house door, plentifully decorated with wood carving, will not shrink or warp, even where the sun shines hottest on it, when it is frequently treated to saturation with wax and oil. Here a plain dosage with linseed oil is sufficient. Varnish, without the addition of turpentine, should never be used, or if used it should be followed by a coat of wax.
III. A good floor wax is composed of 2 parts of wax and 3 parts of Venice turpentine, melted on the water bath, and the mixture applied while still hot, using a pencil, or brush, for the application, and when it has become solid and dry, diligently rubbed, or polished down with a woolen cloth, or with a floor brush, especially made for the purpose.
IV. An emulsion of 5 parts of yellow wax, 2 parts of crude potassium carbonate, and 12 parts of water, boiled together until they assume a milky color and the solids are dissolved, used cold, makes an excellent composition for floors. Any desired color may be given this dressing by stirring in the powdered coloring matter. Use it exactly as described for the first mass.
Gilders’ Wax. For the production of various colorings of gold in fire gilding, the respective places are frequently covered with so-called gilders' wax. These consist of mixtures of various chemicals which have an etching action in the red heat upon the bronze mass, thus causing roughness of unequal depth, as well as through the fact that the composition of the bronze is changed somewhat on the surface, a relief of the gold color being effected in consequence of these two circumstances. The gilding wax is prepared by melting together the finely powdered chemicals with wax according to the following recipes:
|
|
I |
II |
III |
IV |
V |
|
Yellow wax |
32 |
32 |
32 |
96 |
36 |
|
Red chalk |
3 |
24 |
18 |
48 |
18 |
|
Verdigris |
2 |
4 |
18 |
32 |
18 |
|
Burnt alum |
2 |
4 |
|
|
|
|
Burnt borax |
|
|
2 |
1 |
3 |
|
Copper ash |
|
4 |
6 |
20 |
8 |
|
Zinc vitrol |
|
|
|
32 |
18 |
|
Green vitrol |
|
|
|
1 |
6 |
Grafting Wax.
I.
Beeswax 7 parts
Purified rosin 12 parts
Turpentine 3 parts
Rape oil 1 part
Venice turpentine 2.5 parts
Zinc white 2.5 parts
Color yellow with turmeric.
II.
Japan wax 1 part
Yellow wax 3 parts
Rosin 8 parts
Turpentine 4 parts
Hard paraffine 1 part
Suet 3 parts
Venice turpentine 6 parts
Harness Wax.
Oil of turpentine 90 parts
Wax, yellow 9 parts
Prussian blue 1 part
Indigo 0.5 parts
Bone black 5 parts
Dissolve the wax in the oil by aid of a low heat, on a water bath. Mix the remaining ingredients, which must be well powdered, and work up with a portion of the solution of wax. Finally, add the mixture to the solution, and mix thoroughly on the bath. When a homogeneous liquid is obtained, pour into earthen boxes.
Modeling Wax.
I. Yellow wax, 1,000 parts; Venice turpentine, 130 parts; lard, 65 parts; bole, 725 parts. The mixture when still liquid is poured into tepid water and kneaded until a plastic mass is obtained.
II. Summer Modeling Wax. White wax, 20 parts; ordinary turpentine, 4 parts; sesame oil, 1 part; vermilion, 2 parts.
III. Winter Modeling Wax. White wax, 20 parts; ordinary turpentine, 6 parts; sesame oil, 2 parts; vermilion, 2 parts. Preparation same as for Formula I.
Sealing Waxes. The following formulas may be followed for making sealing wax: Take 4 pounds of shellac, 1 pound of Venice turpentine, and 3 pounds of vermilion. Melt the lac in a copper pan suspended over a clear charcoal fire, then add the turpentine slowly to it, and soon afterwards add the vermilion, stirring briskly all the time with a rod in either hand. In forming the round sticks of sealing wax, a certain portion of the mass should be weighed while it is ductile, divided into the desired number of pieces, and then rolled out upon a warm marble slab by means of a smooth wooden block like that used by apothecaries for rolling a mass of pills.
The oval and square sticks of sealing wax are cast in molds, with the above compound, in a state of fusion. The marks of the lines of junction of the mold box may be afterwards removed by holding the sticks over a clear fire, or passing them over a blue gas flame.
Marbled sealing wax is made by mixing
[756]
WAX - WEATHER FORECASTERS
two, three, or more colored kinds together while they are in a semi-fluid state. From the viscidity of the several portions their incorporation is left incomplete, so as to produce the appearance of marbling. Gold sealing wax is made simply by adding gold chrome instead of vermilion into the melted rosins. Wax may be scented by introducing a little essential oil, essence of musk, or other perfume. If 1 part of balsam of Peru be melted along with 99 parts of the sealing-wax composition, an agreeable fragrance will be exhaled in the act of sealing with it. Either lampblack or ivory black serves for the coloring matter of black wax. Sealing wax is often adulterated with rosin, in which case it runs into thin drops at the flame of a candle.
The following mistakes are sometimes made in the manufacture of sealing
wax:
I. Use of filling agents which are too coarsely ground.
II. Excessive use of filling agents.
III. Insufficient binding of the pigments and fillings with a suitable adhesive agent, which causes these bodies to absorb the adhesive power of the gums.
IV. Excessive heating of the mass, caused by improper melting or faulty admixture of the gummy bodies. Turpentine and rosin must be heated before entering the shellac. If this rule is inverted, as is often the case, the shellac sticks to the bottom and burns partly.
Great care must be taken to mix the coloring matter to a paste with spirit or oil of turpentine before adding to the other ingredients. Unless this is done the wax will not be of a regular tint.
Dark Blue Wax. Three ounces Venetian turpentine, 4 ounces shellac, 1 ounce rosin, 1 ounce Prussian blue, 1/2 ounce magnesia.
Green Wax. Two ounces Venetian turpentine, 4 ounces shellac, 1 1/4 ounces rosin, 1/2 ounce chrome yellow, 1/4 ounce Prussian blue, 1 ounce magnesia.
Carmine Red Wax. One ounce Venetian turpentine, 4 ounces shellac, 1 ounce rosin, colophony, 1 1/4 ounces Chinese red, 1 drachm magnesia, with oil of turpentine.
Gold Wax. Four ounces Venetian turpentine, 8 ounces shellac, 14 sheets of genuine leaf gold, 1/2 ounce bronze, 1/2 ounce magnesia, with oil of turpentine.
White Wax.
I. The wax is bleached by exposing to moist air and to the sun, but it must first be prepared in thin sheets or ribbons or in grains. For this purpose it is first washed, to free it from the honey which may adhere, melted, and poured into a tin vessel, whose bottom is perforated with narrow slits. The melted wax falls in a thin stream on a wooden cylinder arranged below and half immersed in cold water. This cylinder is turned, and the wax, rolling round in thin leaves, afterwards falls into the water. To melt it in grains, a vessel is made use of, perforated with small openings, which can be rotated. The wax is projected in grains into the cold water. It is spread on frames of muslin, moistened with water several times a day, and exposed to the sun until the wax assumes a fine white. This whiteness, however, is not perfect. The operation of melting and separating into ribbons or grains must be renewed. Finally, it is melted and flowed into molds. The duration of the bleaching may be abridged by adding to the wax, treated as above, from 1.25 to 1.75 per cent of rectified oil of turpentine, free from rosin. In 6 or 8 days a result will be secured which would otherwise require 5 or 6 weeks.
II. Bleached shellac 28 parts
Venetian turpentine 13 parts
Plaster of Paris 30 parts
WAX FOR BOTTLES:
See Photography.
WAX, BURNING, TRICK:
See Pyrotechnics.
WAXES, DECOMPOSITION OF:
See Oil.
WAX FOR IRONING:
See Laundry Preparations.
WAX FOR LINOLEUM:
See Linoleum.
Weather Forecasters
(See also Hygrometers and Hygroscopes.)
I. It is known that a leaf of blotting paper or a strip of fabric made to change color according to the hygrometric state of the atmosphere has been employed for weather indications in place of a barometer. The following compound is recommended for this purpose: One part of cobalt chloride, 75 parts of nickel oxide, 20 parts of gelatin, and 200 parts
of water. A strip of calico, soaked in this solution, will appear green in fine weather, but when moisture intervenes the color disappears.
[757]
WEATHER FORECASTERS
II.
Copper chloride 1 part
Gelatin 10 parts
Water 100 parts
III. This is a method of making old-fashioned weather glasses containing a liquid that clouds or solidifies under certain atmospheric conditions:
Camphor 2 1/2 drachms
Alcohol 11 drachms
Water 9 drachms
Saltpeter 38 grains
Sal ammoniac 38 grains
Dissolve the camphor in the alcohol and the salts in the water and mix the solutions together. Pour in test tubes, cover with wax after corking and make a hole through the cork with a red-hot needle, or draw out the tube until only a pin hole remains. When the camphor, etc., appear soft and powdery, and almost filling the tube, rain with south or southwest winds may be expected; when crystalline, north, northeast, or northwest winds, with fine weather, may be expected; when a portion crystallizes on one side of the tube, wind may be expected from that direction. Fine weather: The substance remains entirely at bottom of tube and the liquid perfectly clear. Coming rain: Substance will rise gradually, liquid will be very clear, with a small star in motion. A coming storm or very high wind: Substance partly at top of tube, and of a leaflike form, liquid very heavy and in a fermenting state. These effects are noticeable 24 hours before the change sets in. In winter: Generally the substance lies higher in the tube. Snow or white frost: Substance very white and small stars in motion. Summer weather: The substance will lie quite low. The substance will lie closer to the tube on the opposite side to the quarter from which the storm is coming. The instrument is nothing more than a scientific toy.
WEATHERPROOFING:
See Paints.
WEED KILLERS:
See Disinfectants.
Weights and Measures
INTERNATIONAL ATOMIC WEIGHTS.
The International Committee on Atomic Weights have presented this table as corrected:
|
|
|
O=16 |
H=1 |
|
Aluminum Antimony Argon Arsenic Barium Bismuth Boron Bromine Cadmium Caesium Calcium Carbon Cerium Chlorine Chromium Cobalt Columbium Copper Erbium Fluorine Gadolinium Gallium Germanium Glucinum Gold Helium Hydrogen Indium Iodine Iridium Iron Krypton Lanthanum Lead Lithium Magnesium Manganese Mercury Molybdenum Neodymium Neon Nickel Nitrogen Osmium Oxygen Palladium Phosphorus Platinum Potassium Praseodymium Radium Rhodium Rubidium Ruthenium Samarium Scandium Selenium Silicon Silver Sodium Strontium Sulphur Tantalum Tellurium Terbium Thallium Thorium Thulium Tin Titanium Tungsten Uranium Vanadium Xenon Ytterbium Yttrium Zinc Zirconium |
Al Sb A As Ba Bi B Br Cd Cs Ca C Ce Cl Cr Co Cb Cu Er F Gd Ga Ge Gl Au He H In I Ir Fe Kr La Pb Li Mg Mn Hg Mo Nd Ne Ni N Os O Pd P Pt K Pr Ra Rh Rb Ru Sm Sc Se Si Ag Na Sr S Ta Te Tb Tl Th Tm Sn Ti W U V Xe Yb Yt Zn Zr |
27.1 120.2 39.9 75 137.4 208.5 11 79.96 112.4 132.9 40.1 12 140.25 35.45 52.1 59 94 63.6 166 19 156 70 72.5 9.1 197.2 4 1.008 115 126.97 193 55.9 81.8 138.9 206.9 7.03 24.36 55 200 96 143.6 20 58.7 14.04 191 16 106.5 31 194.8 39.15 140.5 225 103 85.5 101.7 150.3 44.1 79.2 28.4 87.6 23.05 87.6 32.06 183 127.6 160 204.1 232.5 171 119 48.1 184 238.5 51.2 128 173 89 65.4 90.6 |
26.9 119.3 39.6 74.4 136.4 206.9 10.9 79.36 111.6 131.9 39.7 11.91 139.2 35.18 51.7 58.55 93.3 63.1 164.8 18.9 154.8 69.5 72 9.03 195.7 4 1 114.1 126.01 191.5 55.5 81.2 137.9 205.35 6.98 24.18 54.6 198.5 95.3 142.5 19.9 58.3 13.93 189.6 15.88 105.7 30.77 193.3 38.85 139.4 223.3 102.2 8.9 100.9 149.2 43.8 78.6 28.2 107.11 22.88 86.94 31.82 181.6 126.6 158.8 202.6 230.8 169.7 118.1 47.7 182.6 236.7 50.8 127 171.7 88.3 64.9 89.9 |
UNITED STATES WEIGHTS AND MEASURES
(According to existing standards)
LINEAL

SURFACE LAND

VOLUME LIQUID
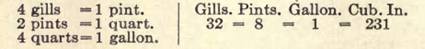
FLUID MEASURE

TROY WEIGHT
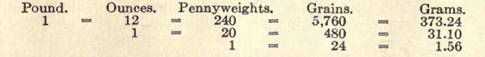
APOTHECARIES' WEIGHT

AVOIRDUPOIS WEIGHT
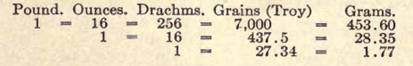
ENGLISH WEIGHTS AND MEASURES

[759]
WEIGHTS AND MEASURES
METRIC SYSTEM OF WEIGHTS AND MEASURES
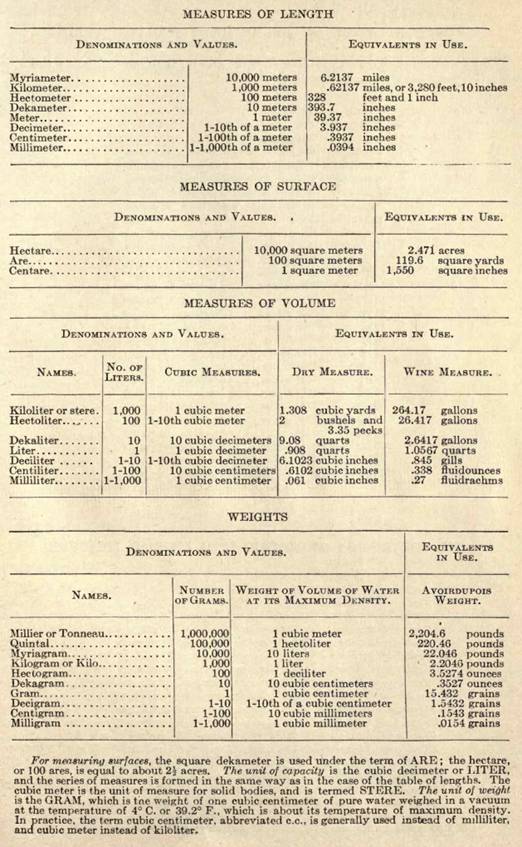
[760]
WEIGHTS AND MEASURES
THE CONVERSION OF METRIC INTO ENGLISH WEIGHT
The following table, which contains no error greater than one-tenth of a grain, will suffice for most practical purposes:
1 gram = 15 2/5 grains
2 grams = 30 4/5 grains
3 grams = 46 1/5 grains
4 grams = 61 4/5 grains, or 1 drachm, 1 4/5 grains
5 grams = 77 1/5 grains, or 1 drachm, 17 1/5 grains
6 grams = 92 3/5 grains, or 1 drachm, 32 3/5 grains
7 grams = 108 grains, or 1 drachm, 48 grains
8 grams = 123 2/5 grains, or 2 drachms, 3 2/5 grains
9 grams = 138 4/5 grains, or 2 drachms, 18 4/5 grains
10 grams = 154 2/5 grains, or 2 drachms, 34 2/5 grains
11 grams = 169 4/5 grains, or 2 drachms, 49 4/5 grains
12 grams = 185 1/5 grains, or 3 drachms, 5 1/5 grains
13 grams = 200 3/5 grains, or 3 drachms, 20 3/5 grains
14 grams = 216 grains, or 3 drachms, 36 grains
15 grams = 23 2/5 grains, or 3 drachms, 5 2/5 grains
16 grams = 247 grains, or 4 drachms, 7 grains
17 grams = 262 2/5 grains, or 4 drachms, 22 2/5 grains
18 grams = 277 4/5 grains, or 4 drachms, 37 4/5 grains
19 grams = 293 1/5 grains, or 4 drachms, 53 1/5 grains
20 grams = 308 3/5 grains, or 5 drachms, 8 3/5 grains
30 grams = 463 grains, or 7 drachms, 43 grains
40 grams = 617 1/5 grains, or 1 drachms, 17 1/5 grains
50 grams = 771 3/5 grains, or 12 drachms, 51 3/5 grains
60 grams = 926 grains, or 15 drachms, 26 grains
70 grams = 1,080 1/5 grains, or 18 drachms, 0 1/5 grains
80 grams = 1,234 3/5 grains, or 20 drachms, 34 3/5 grains
90 grams = 1,389 grains, or 23 drachms, 9 grains
100 grams= 1,543 1/5 grains, or 25 drachms, 43 1/5 grains
1,000 grams = 1 kilogram =32 ounces, 1 drachm, 12 2/5 grains
THE CONVERSION OF METRIC INTO ENGLISH MEASURE
1 cubic centimeter = 17 minims
2 cubic centimeters = 34 minims
3 cubic centimeters = 51 minims
4 cubic centimeters = 68 minims, or 1 drachm, 8 minims
5 cubic centimeters = 85 minims, or 1 drachm, 25 minims
6 cubic centimeters = 101 minims, or 1 drachm, 41 minims
7 cubic centimeters = 118 minims, or 1 drachm, 58 minims
8 cubic centimeters = 135 minims, or 2 drachms, 15 minims
9 cubic centimeters = 152 minims, or 2 drachms, 32 minims
10 cubic centimeters = 169 minims, or 2 drachms, 49 minims
20 cubic centimeters = 338 minims, or 5 drachms, 38 minims
30 cubic centimeters = 507 minims, or 1 ounce, drachm, 27 minims
40 cubic centimeters = 676 minims, or 1 ounce, 3 drachms, 16 minims
50 cubic centimeters = 845 minims, or 1 ounce, 6 drachms, 5 minima
60 cubic centimeters = 1,014 minims, or 2 ounces, drachms, 54 minims
70 cubic centimeters = 1,183 minims, or 2 ounces, 3 drachms, 43 minims
80 cubic centimeters = 1,352 minims, or 2 ounces, 6 drachms, 32 minims
90 cubic centimeters = 1,521 minims, or 3 ounces, 1 drachm, 21 minims
100 cubic centimeters = 1,690 minims, or 3 ounces, 4 drachms, 10 minims
1,000 cubic centimeters = 1 liter = 34 fluidounces nearly, or 2 1/8[?] pints
[761]
WELDING POWDERS - WHITEWASH
WELDING POWDERS.
See also Steel.
Powder to Weld Wrought Iron at Palered Heat with Wrought Iron.
I. Borax, 1 part (by weight); sal ammoniac, 1/2 part; water, 1/2 part. These ingredients are boiled with constant stirring until the mass is stiff; then it is allowed to harden over the fire. Upon cooling, the mass is rubbed up into a powder and mixed with one-third wrought-iron filings free from rust. When the iron has reached red heat, this powder is sprinkled on the parts to be welded, and after it has liquefied, a few blows are sufficient to unite the pieces.
II. Borax, 2 parts; wrought-iron filings, free from rust, 2 parts; sal ammoniac, 1 part. These pulverized parts are moistened with copaiba balsam and made into a paste, then slowly dried over a fire and again powdered. The application is the same as for Formula I.
Welding Powder to Weld Steel on Wrought Iron at Pale Red Heat. Borax,
3 parts; potassium cyanide, 2 parts; Berlin blue, 1-100 part. These substances are powdered well, moistened with water; next they are boiled with constant stirring until stiff; then dry over a fire. Upon cooling, the mass is finely pulverized and mixed with 1 part of wrought-iron filings, free from rust. This powder is sprinkled repeatedly upon the hot pieces, and after it has burned in the welding is taken in hand.
WHEEL GREASE:
See Lubricants.
WHETSTONES.
To make artificial whetstones, take gelatin of good quality, dissolve it in equal weight of water, operating in almost complete darkness, and add 1 1/2 per cent of bichromate of potash, previously dissolved. Next take about 9 times the weight of the gelatin employed of very fine emery or fine powdered gun stone, which is mixed intimately with the gelatinized solution. The paste thus obtained is molded into the desired shape, taking care to exercise an energetic pressure in order to consolidate the mass. Finally dry by exposure to the sun.
WHITING:
To Form Masses of Whiting. Mix the whiting into a stiff paste with water, and the mass will retain its coherence when dry.
Whitewash
(See also Paint.)
Wash the ceiling by wetting it twice with water, laying on as much as can well be floated on, then rub the old color up with a stumpy brush and wipe off with a large sponge. Stop all cracks with whiting and plaster of Paris. When dry, claricole with size and a little of the whitewash when this is dry. If very much stained, paint those parts with turps, color, and, if necessary, claricole again. To make the whitewash, take a dozen pounds of whiting (in large balls), break them up in a pail, and cover with water to soak. During this time melt over a slow fire 4 pounds common size, and at the same time, with a palette knife or small trowel, rub up fine about a dessertspoonful of blue-black with water to a fine paste; then pour the water off the top of the whiting and with a stick stir in the black; when well mixed, stir in the melted size and strain. When cold, it is fit for use. If the jelly is too stiff for use, beat it up well and add a little cold water. Commence whitewashing over the window and so work from the light. Distemper color of any tint may be made by using any other color instead of the blue-black as ocher, chrome, Dutch pink, raw sienna for yellows and buff; Venetian red, burnt sienna, Indian red or purple brown for reds; celestial blue, ultramarine, indigo for blues; red and blue for purple, gray or lavender; red lead and chrome for orange; Brunswick green for greens.
Ox blood in lime paint is an excellent binding agent for the lime, as it is chiefly composed of albumin, which, like casein or milk, is capable of transforming the lime into casein paint. But the ox blood must be mixed in the lime paint; to use it separately is useless, if not harmful. Whitewashing rough mortar-plastering to saturation is very practical, as it closes all the pores and small holes.
A formula used by the United States Government in making whitewash for light-houses and other public buildings is as follows:
Unslaked lime 2 pecks
Common salt 1 peck
Rice flour 3 pounds
Spanish whiting 1/2 pound
Glue (clean and white) 1 pound
Water a sufficient quantity
Slake the lime in a vessel of about 10 gallons capacity; cover it, strain, and add
[762]
WHITEWASH - WINES AND LIQUORS
the salt previously dissolved in warm water. Boii the rice flour in water; soak the glue in water and dissolve on a water bath, and add both, together with the whiting and 5 gallons of hot water to the mixture, stirring all well together. Cover to protect from dirt, and let it stand for a few days, when it will be ready for use. It is to be applied hot, and for that reason should be used from a kettle over a portable furnace.
To Soften Old Whitewash. Wet the whitewash thoroughly with a wash made
of 1 pound of potash dissolved in 10 quarts of water.
WHITEWASH, TO REMOVE:
See Cleaning Preparations and Methods.
WHITE METAL:
See Alloys.
WINDOW CLEANING COMPOUND:
See Cleaning Compounds.
WINDOW DISPLAY:
See also Sponges.
An attractive window display for stores can be prepared as follows:
In a wide-mouth jar put some sand, say, about 6 inches in depth. Make a mixture of equal parts of aluminum sulphate, copper sulphate, and iron sulphate, coarsely powdered, and strew it over the surface of the sand. Over this layer gently pour a solution of sodium silicate, dissolved in 3 parts of hot water, taking care not to disturb the layer of sulphates. In about a week or 10 days the surface will be covered with crystals of different colors, being silicates of different metals employed. Now take some pure water and let it run into the vessel by a small tube, using a little more of it than you used of the water-glass solution. This will displace the water-glass solution, and a fresh crop of crystals will come in the silicates, and makes, when properly done, a pretty scene. Take care in pouring in the water to let the point of the tube be so arranged as not to disturb the crop of silicates.
WINDOW PERFUME.
In Paris an apparatus has been introduced consisting of a small tube which is attached lengthwise on the exterior of the shop windows. Through numerous little holes a warm, lightly perfumed current of air is passed, which pleasantly tickles the olfactory nerves of the looker-on and at the same time keeps the panes clear and clean, so that the goods exhibited present the best possible appearance.
WINDOW POLISHES:
See Polishes.
WINDOWS, FROSTED:
See Glass.
WINDOWS, TO PREVENT DIMMING OF:
See Glass.
Wines and Liquors
BITTERS.
Bitters, as the name indicates, are merely tinctures of bitter roots and barks, with the addition of spices to flavor, and depend for their effect upon their tonic action on the stomach. Taken too frequently, however, they may do harm, by overstimulating the digestive organs.
The recipes for some of these preparations run to great lengths, one for Angostura bitters containing no fewer than 28 ingredients. A very good article, however, may be made without all this elaboration. The following, for instance, make a very good preparation:
Gentian root (sliced) 12 ounces
Cinnamon bark 10 ounces
Caraway seeds 10 ounces
Juniper berries 2 ounces
Cloves 1 ounce
Alcohol, 90 per cent 7 pints
Macerate for a week; strain, press out, and filter, then add
Capillaire 1 1/4 pints
Water to make up 2 1/2 gallons
Strength about 45 u.p.
Still another formula calls for Angostura bark, 2 1/2 ounces; gentian root, 1 ounce; cardamom seeds, 1/2 ounce; Turkey rhubarb, 1/2 ounce; orange peel, 4 ounces; caraways, 1/2 ounce; cinnamon bark, 1/2 ounce; cloves, 1/4 ounce.
Brandy Bitters.
Sliced gentian root 3 pounds
Dried orange peel 2 pounds
Cardamom seed 1 pound
Bruised cinnamon 1/2 pound
Cochineal 2 ounces
Brandy 10 pints
Macerate for 14 days and strain.
Hostetter's Bitters.
Calamus root 1 pound
Orange peel 1 pound
Peruvian bark 1 pound
Gentian root 1 pound
[763]
WINES AND LIQUORS
Calumba root 1 pound
Rhubarb root 4 ounces
Cinnamon bark 2 ounces
Cloves 1 ounce
Diluted alcohol 2 gallons
Water 1 gallon
Sugar 1 pound
Macerate together for 2 weeks.
CORDIALS.
Cordials, according to the Spatula, are flavored liquors containing from 40 to 50 per cent of alcohol (from 52 to 64 fluidounces to each gallon) and from 20 to 25 per cent of sugar (from 25 to 32 ounces avoirdupois to each gallon).
Cordials, while used in this country to some degree, have their greatest consumption in foreign lands, especially in France and Germany.
Usually such mixtures as these are clarified or "fined" only with considerable difficulty, as the finally divided particles of oil pass easily through the pores of the filter paper. Purified talcum will be found to be an excellent clarifying medium; it should be agitated with the liquid and the liquid then passed through a thoroughly wetted filter. The filtrate should be returned again and again to the filter until it filters perfectly bright. Purified talcum being chemically inert is superior to magnesium carbonate and other substances which are recommended for this purpose.
When the filtering process is completed the liquids should at once be put into suitable bottles which should be filled and tightly corked and sealed. Wrap the bottles in paper and store away, laying the bottles on their sides in a moderately warm place. A shelf near the ceiling is a good place. Warmth and age improve the beverages, as it appears to more perfectly blend the flavors, so that the older the liquor becomes the better it is. These liquids must never be kept in a cold place, as the cold might cause the volatile oils to separate.
The following formulas are for the production of cordials of the best quality, and therefore only the very best of materials should be used; the essential oils should be of unquestionable quality and strictly fresh, while the alcohol must be free from fusel oil, the water distilled, and the sugar white, free from bluing, and if liquors of any kind should be called for in any formula only the very best should be used. The oils and other flavoring substances should be dissolved in the alcohol and the sugar in the water. Then mix the two solutions and filter clear.
Alkermes Cordial.
Mace 1 1/2 avoirdupois ounces
Ceylon cinnamon 1 1/8 avoirdupois ounces
Cloves 3/4 avoirdupois ounce
Rose water (best) 6 fluidounces
Sugar 28 avoirdupois ounces
Deodorized alcohol 52 fluidounces
Distilled water, q.s 1 gallon
Reduce the mace, cinnamon, and cloves to a coarse powder macerate with the alcohol for several days, agitating occasionally, then add the remaining ingredients, and filter clear.
Anise Cordial.
Anethol 7 fluidrachms
Oil of fennel seed 80 minims
Oil of bitter almonds 16 drops
Deodorized alcohol 8 pints
Simple syrup 5 pints
Distilled water, q.s. 16 pints
Mix the oils and anethol with the alcohol and the syrup with the water; mix the two and filter clear, as directed.
Blackberry Cordial. This beverage is usually misnamed "blackberry brandy" or "blackberry wine." This latter belongs only to wines obtained by the fermentation of the blackberry juice. When this is distilled then a true blackberry brandy is obtained, just as ordinary brandy is obtained by distilling ordinary wines.
The name is frequently applied to a preparation containing blackberry root often combined with other astringents, but the true blackberry cordial is made according to the formulas given herewith. Most of these mention brandy, and this article should be good and fusel free, or it may be replaced by good whisky, or even by diluted alcohol, depending on whether a high-priced or cheap cordial is desired.
I. Fresh blackberry juice, 3 pints; sugar, 7 1/2 ounces; water, 30 fluidounces; brandy, 7 1/2 pints; oil of cloves, 3 drops; oil of cinnamon, 3 drops; alcohol, 6 fluidrachms. Dissolve the sugar in the
water and juice, then add the liquor. Dissolve the oils in the alcohol and add 1/2 to the first solution, and if not sufficiently flavored add more of the second solution. Then filter.
II. Fresh blackberry juice, 4 pints; powdered nutmeg (fresh), 1 ounce; powdered cinnamon (fresh), 1 ounce; powdered pimento (fresh), 1/2 ounce; powdered cloves
[764]
WINES AND LIQUORS
(fresh), 1/2 ounce; brandy, 2 1/2 pints; sugar, 2 1/2 pounds. Macerate the spices in the brandy for several days. Dissolve the sugar in the juice and mix and filter clear.
Cherry Cordials.
I.
Oil of bitter almonds 8 drops
Oil of cinnamon 1 drop
Oil of cloves 1 drop
Acetic ether 12 drops
Ceuanthic ether 1 drop
Vanilla extract 1 drachm
Alcohol 3 pints
Sugar 3 pounds
Cherry juice 20 ounces
Distilled water, q.s. 1 gallon
The oils, ethers, and extracts must be dissolved in the alcohol, the sugar in part of the water, then mix, add the juice and filter clear. When the juice is not sufficiently sour, add a small amount of solution of citric acid. To color, use caramel.
II.
Vanilla extract 10 drops
Oil of cinnamon 10 drops
Oil of bitter almonds 10 drops
Oil of cloves 3 drops
Oil of nutmeg 3 drops
Alcohol 2 1/2 pints
Cherry juice 2 1/2 pints
Simple syrup 3 pints
Dissolve the oils in the alcohol, then add the other ingredients and filter clear. It is better to make this cordial during the cherry season so as to obtain the fresh expressed juice of the cherry.
Curacoa Cordials.
I.
Curacoa orange peel 6 ounces
Cinnamon 3/4 ounce
Mace 2 1/2 drachms
Alcohol 3 1/2 pints
Water 4 1/2 pints
Sugar 12 ounces
Mix the first three ingredients and reduce them to a coarse powder, then mix with the alcohol and 4 pints of water and macerate for 8 days with an occasional agitation, express, add the sugar and enough water to make a gallon of finished product. Filter clear.
II.
Curacoa or bitter orange peel 2 ounces
Cloves 80 grains
Cinnamon 80 grains
Cochineal 60 grains
Oil of orange (best) 1 drachm
Orange-flower water 1/2 pint
Holland gin 1 pint
Alcohol 2 pints
Sugar 3 pints
Water, q.s. 1 gallon
Reduce the solids to a coarse powder, add the alcohol and macerate 3 days. Then add the oil, gin, and 3 pints of water and continue the maceration for 8 days more, agitating once a day, strain and add sugar dissolved in balance of the water. Then add the orange-flower water and filter.
Kola Cordial.
Kola nuts, roasted and powdered 7 ounces
Cochineal powder 30 grains
Extract of vanilla 3 drachms
Arrac 3 ounces
Sugar 7 pounds
Alcohol 6 pints
Water, distilled 6 pints
Macerate kola and cochineal with alcohol for 10 days, agitate daily, dd
arrac, vanilla, and sugar dissolved in water. Filter.
Kiimmel Cordials.
I.
Oil of caraway 30 drops
Oil of peppermint 3 drops
Oil of lemon 3 drops
Acetic ether 30 drops
Spirit of nitrous ether 30 drops
Sugar 72 ounces
Alcohol 96 ounces
Water 96 ounces
Dissolve the oils and ethers in the alcohol, and the sugar in the water. Mix and filter.
II.
Oil of caraway 20 drops
Oil of sweet fennel 2 drops
Oil of cinnamon 1 drop
Sugar 14 ounces
Alcohol 2 pints
Water 4 pints
Prepare as in Formula I.
Orange Cordials. Many of the preparations sold under this name are not really orange cordials, but are varying mixtures of uncertain composition, possibly flavored with orange. The following are made by the use of oranges:
I.
Sugar 8 avoirdupois pounds
Water 2 3/4 gallons
Oranges 15
Dissolve the sugar in the water by the aid of a gentle heat, express the oranges, add the juice and rinds to the syrup, put the mixture into a cask, keep the whole in a warm place for 3 or 4 days, stirring frequently, then close the cask, set aside in a cool cellar and draw off the clear liquid.
II. Express the juice from sweet oranges, add water equal to the volume
[765]
WINES AND LIQUORS
of juice obtained, and macerate the expressed oranges with the juice and water for about 12 hours. For each gallon of juice, add 1 pound of granulated sugar, grape sugar, or glucose, put the whole into a suitable vessel, covering to exclude the dust, place in a warm location until fermentation is completed, draw off the clear liquid, and preserve in well-stoppered stout bottles in a cool place.
III. Orange wine suitable for "soda" purposes may be prepared by mixing 3 fluidounces of orange essence with 13 fluidounces of sweet Catawba or other mild wine. Some syrup may be added to this if desired.
Rose Cordial.
Oil of rose, very best 3 drops
Palmarosa oil 3 drops
Sugar 28 ounces
Alcohol 52 ounces
Distilled water, q.s. 8 pints
Dissolve the sugar in the water and the oils in the alcohol; mix the solutions, color a rose tint, and filter clear.
Spearmint Cordial.
Oil of spearmint 30 drops
Sugar 28 ounces
Alcohol 52 ounces
Distilled water, q. s 8 pints
Dissolve the sugar in the water and the
oil in the alcohol; mix the two solutions,
color green, and filter clear.
Absinthe.
I.
Oil of wormwood 96 drops
Oil of star anise 72 drops
Oil of aniseed 48 drops
Oil of coriander 48 drops
Oil of fennel, pure 48 drops
Oil of angelica root 24 drops
Oil of thyme 24 drops
Alcohol (pure) 162 fluidounces
Distilled water 30 fluidounces
Dissolve the oils in the alcohol, add the water, color green, and filter clear.
II.
Oil of wormwood 36 drops
Oil of orange peel 30 drops
Oil of star anise 12 drops
Oil of neroli petate 5 drops
Fresh oil of lemon 9 drops
Acetic ether 24 drops
Sugar 30 avoirdupois ounces
Alcohol, deodorized 90 fluidounces
Distilled water 78 fluidounces
Dissolve the oils and ether in the alcohol and the sugar in the water; then mix thoroughly, color green, and filter clear.
DETANNATING WINE.
According to Caspari, the presence of appreciable quantities of tannin in wine is decidedly objectionable if the wine is to be used in connection with iron and other metallic salts; moreover, tannin is incompatible with alkaloids, and hence wine not deprived of its tannin should never be used as a menstruum for alkaloidal drugs. The process of freeing wines from tannin is termed detannation, and is a very simple operation. The easiest plan is to add 1/2 ounce of gelatin in number 40 or number 60 powder to 1 gallon of the wine, to agitate occasionally during 24 or 48 hours, and then to filter. The operation is preferably carried out during cold weather or in a cold apartment, as heat will cause the gelatin to dissolve, and the maceration must be continued until a small portion of the wine mixed with a few drops of ferric chloride solution shows no darkening of color. Gelatin in large pieces is not suitable, especially with wines containing much tannin, since the newly formed tannate of gelatin will be deposited on the surface and prevent further intimate contact of the gelatin with the wine. Formerly freshly prepared ferric hydroxide was much employed for detannating wine, but the chief objection to its use was due to the fact that some iron invariably was taken up by the acid present in the wine; moreover, the process was more tedious than in the case of gelatin. As the removal of tannin from wine in no way interferes with its qualitv alcoholic strength and aroma remaining the same, and only coloring matter being lost a supply of detannated wine should be kept on hand, for it requires very little more labor to detannate a gallon than a pint.
If ferric hydroxide is to be used, it must be freshly prepared, and a convenient quantity then be added to the wine about 8 ounces of the expressed, but moist, precipitate to a gallon.
PREVENTION OF FERMENTATION.
Fermentation may be prevented in either of two ways:
(1) By chemical methods, which consist in the addition of germ poisons or antiseptics, which either kill the germs or prevent their growth. Of these the principal ones used are salicylic, sulphurous, boracic, and benzoic acids, formalin, fluorides, and saccharine. As these substances are generally regarded as adulterants and injurious, their use is not recommended.
(2) The germs are either removed by
[766]
WINES AND LIQUORS
some mechanical means such as a filtering or a centrifugal apparatus, or they are destroyed by heat or electricity. Heat has so far been found the most practical.
When a liquid is heated to a sufficiently high temperature all organisms in it are killed. The degree of heat required, however, differs not only with the particular kind of organism, but also with the liquid in which it is held. Time is also a factor. An organism may not be killed if heated to a high temperature and quickly cooled. If, however, the temperature is kept at the same high degree for some time, it will be killed. It must also be borne in mind that fungi, including yeasts, exist in the growing and the resting states, the latter being much more resistant than the former. One characteristic of the fungi and their spores is their great resistance to heat when dry. In this state they can be heated to 212º F. without being killed. The spores of the common mold are even more resistant. This should be well considered in sterilizing bottles and corks, which should be steamed to 240º F. for at least 15 minutes.
Practical tests so far made indicate that grape juice can be safely sterilized at from 165º to 176º F. At this temperature the flavor is hardly changed, while at a temperature much above 200º F. it is. This is an important point, as the flavor and quality of the product depend on it.
Use only clean, sound, well-ripened, but not over-ripe grapes. If an ordinary cider mill is at hand, it may be used for crushing and pressing, or the grapes may be crushed and pressed with the hands. If a light-colored juice is desired, put the crushed grapes in a cleanly washed cloth sack and tie up. Then either hang up securely and twist it or let two persons take hold, one on each end of the sack and twist until the greater part of the juice is expressed. Next gradually heat the juice in a double boiler or a large stone jar in a pan of hot water, so that the juice does not come in direct contact with the fire at a temperature of 180º to 200º F., never above 200º F. It is best to use a thermometer, but if there be none at hand heat the juice until it steams, but do not allow it to boil. Put it in a glass or enameled vessel to settle for 24 hours; carefully drain the juice from the sediment, and run it through several thicknesses of clean flannel, or a conic filter made from woolen cloth or felt may be used. This filter is fixed to a hoop of iron, which can be suspended wherever necessary. After this fill into clean bottles. Do not fill entirely, but leave room for the liquid to expand when again heated. Fit a thin board over the bottom of an ordinary wash boiler, set the filled bottles (ordinary glass fruit jars are just as good) in it, fill in with water around the bottles to within about an inch of the tops, and gradually heat until it is about to simmer. Then take the bottles out and cork or seal immediately. It is a good idea to take the further precaution of sealing the corks over with sealing wax or paraffine to prevent mold germs from entering through the corks. Should it be desired to make red juice, heat the crushed grapes to not above 200º F., strain through a clean cloth or drip bag (no pressure should be used), set away to cool and settle, and proceed the same as with light-colored juice. Many people do not even go to the trouble of letting the juice settle after straining it, but reheat and seal it up immediately, simply setting the vessel away in a cool place in an upright position where they will be undisturbed. The juice is thus allowed to settle, and when wanted for use the clear juice is simply taken off the sediment. Any person familiar with the process of canning fruit can also preserve grape juice, for the principles involved are identical.
One of the leading defects so far found in unfermented juice is that much of it is not clear, a condition which very much detracts from its otherwise attractive appearance, and due to two causes already alluded to. Either the final sterilization in bottles has been at a higher temperature than the preceding one, or the juice has not been properly filtered or has not been filtered at all. In other cases the juice has been sterilized at such a high temperature that it has a disagreeable scorched taste. It should be remembered that attempts to sterilize at a temperature above 195º F. are dangerous so far as the flavor of the finished product is concerned.
Another serious mistake is sometimes made by putting the juice into bottles so large that much of it becomes spoiled before it is used after the bottles are opened. Unfermented grape juice properly made and bottled will keep indefinitely, if it is not exposed to the atmosphere or mold germs; but when a bottle is once opened it should, like canned goods, be used as soon as possible to keep from spoiling.
Another method of making unfermented grape juice, which is often re-
[767]
WINES AND LIQUORS
sorted to where a sufficiently large quantity is made at one time, consists in this:
Take a clean keg or barrel (one that has previously been made sweet). Lay this upon a skid consisting of two scantlings or pieces of timber of perhaps 20 feet long, in such a manner as to make a runway. Then take a sulphur match, made by dipping strips of clean muslin about 1 inch wide and 10 inches long into melted brimstone, cool it and attach it to a piece of wire fastened in the lower end of a bung and bent over at the end, so as to form a hook. Light the match and by means of the wire suspend it in the barrel, bung the barrel up tight, and allow it to burn as long as it will. Repeat this until fresh sulphur matches will no longer burn in the barrel.
Then take enough fresh grape juice to fill the barrel one-third full, bung up tight, roll and agitate violently on the skid for a few minutes. Next burn more sulphur matches in it until no more will burn, fill in more juice until the barrel is about two-thirds full; agitate and roll again. Repeat the burning process as before, after which fill the barrel completely with grape juice and roll. The barrehshould then be bunged tightly and stored in a cool place with the bung up, and so secured that the package cannot be shaken. In the course of a few weeks the juice will have become clear and can then be racked off and filled into bottles or jars direct, sterilized, and corked or sealed up ready for use. By this method, however, unless skillfully handled, the juice is apt to have a slight taste of the sulphur.
The following are the component parts of a California and a Concord unfermented grape juice:
|
|
Concord Per Cent |
California Per Cent |
|
Solid contents |
20.37 |
20.60 |
|
Total acids (as tartaric) |
.663 |
.53 |
|
Volatile acids |
.023 |
.03 |
|
Grape sugar |
18.54 |
19.15 |
|
Free tartaric acids |
.025 |
.07 |
|
Ash |
.255 |
.19 |
|
Phosphoric acids |
.027 |
.04 |
|
Cream of tartar |
.55 |
.59 |
This table is interesting in so far that the California unfermented grape juice was made from Viniferas or foreign varieties, whereas the Concord was a Labruska or one of the American sorts. The difference in taste and smell is even more pronounced than the analysis would indicate.
Small quantities of grape juice may be preserved in bottles. Fruit is likely to be dusty and to be soiled in other ways, and grapes, like other fruits', should be well washed before using. Leaves or other extraneous matter should also be removed. The juice is obtained by moderate pressure in an ordinary screw press, and strained through felt. By gently heating, the albuminous matter is coagulated and may be skimmed off, and further clarification may be effected by filtering through paper, but such filtration must be done as rapidly as possible, using a number of filters and excluding the air as much as possible.
The juice so obtained may be preserved by sterilization, in the following manner: Put the juice in the bottles in which it is to be kept, filling them very nearly full; place the bottles, unstoppered, in a kettle filled with cold water, so arranging them on a wooden perforated "false bottom" or other like contrivance as to prevent their immediate contact with the metal, this preventing unequal heating and possible fracture. Now heat the water, gradually raising the temperature to the boiling point, and maintain at that until the juice attains a boiling temperature; then close the bottles with perfectly fitting corks, which have been kept immersed in boiling water for a short time before use.
The corks should not be fastened in any way, for, if the sterilization is not complete, fermentation and consequent explosion of the bottle may occur unless the cork should be forced out.
If the juice is to be used for syrup, as for use at the soda fountain, the best method is to make a concentrated syrup at once, using about 2 pounds of refined sugar to 1 pint of juice, dissolving by a gentle heat. This syrup may be made by simple agitation without heat; and a finer flavor thus results, but its keeping quality would be uncertain.
The juices found in the market are frequently preserved by means of antiseptics, but so far none have been proposed for this purpose which can be considered entirely wholesome. Physiological experiments have shown that while bodies suited for this purpose may be apparently without bad effect at first, their repeated ingestion is likely to cause gastric disturbance.
SPARKLING WINES.
An apparatus for converting still into foaming wines, and doing this efficiently, simply, and rapidly, consists of a vertical steel tube, which turns on an axis, and
[768]
WINES AND LIQUORS
bears several adjustable glass globes that are in connection with each other by means of distributing valves, the latter being of silver-plated bronze. The glass globes serve as containers for carbonic acid, and are kept supplied with this gas from a cylinder connected therewith.
The wine to be impregnated with the acid is taken from a cask, through a special tube, which also produces a light pressure of carbonic acid on the cask, the object of which is to prevent the access of atmospheric air to the wine within, and, besides, to cause the liquid to pass into the bottle without jar or stroke. The bottles stand under the distributing valves, or levers, placed above and below them. Now, if the cock, by means of which the glass bulbs and the bottles are brought into connection, is slightly opened, and the desired lever is put in action, the carbonic acid at once forces the air out of the bottles, and sterilizes them. The upper bottles are now gradually filled. The whole apparatus, including the filled bottles, is now tilted over, and the wine, of its own weight, flows through collectors filled with carbonic acid, and passes, impregnated with the gas, into other bottles placed below. Each bottle is filled in course, the time required for each being some 45 seconds. The saturation of the liquid with carbonic acid is so complete and plentiful that there is no need of hurry in corking.
By means of this apparatus any desired still wine is at once converted into a sparkling one, preserving at the same time its own peculiarities of taste, bouquet, etc. The apparatus may be used equally well upon fruit juices, milk, and, in fact, any kind of liquid, its extreme simplicity permitting of easy and rapid cleansing.
ARTIFICIAL FRENCH BRANDY.
I. The following is Eugene Dieterich's formula for Spiritus vini Gallici artificialis:
Tincture of gall-apples 10 parts
Aromatic tincture 5 parts
Purified wood vinegar 5 parts
Spirit of nitrous ether 10 parts
Acetic ether 1 part
Alcohol, 68 per cent 570 parts
Distilled water 400 parts
Mix, adding the water last, let stand for several days, then filter.
II. The Münchener Apotheker Verein has adopted the following formula for the same thing:
Acetic acid, dilute, 90 per cent 4 parts
Acetic ether 4 parts
Tincture aromatic 40 parts
Cognac essence 40 parts
Spirit of nitrous ether 20 parts
Alcohol, 90 per cent 5,000 parts
Water, distilled 2,500 parts
Add the acids, ethers, etc., to the alcohol, and finally add the water. Let stand several days, and, if necessary, filter.
III. The Berlin Apothecaries have adopted the following as a magistral
formula:
Aromatic tincture 4 parts
Spirit of nitrous ether 5 parts
Alcohol, 90 per cent 1,000 parts
Distilled water,
quantity sufficient to make 2,000 parts
Mix the tincture and ether with the alcohol, add the water and for every ounce add one drop of tincture of rhatany.
Of these formulas the first is to be preferred as a close imitation of the taste of the genuine article. To imitate the color use burnt sugar.
LIQUEURS.
Many are familiar with the properties of liqueurs but believe them to be very complex and even mysterious compounds. This is, of course, due to the fact that the formulas are of foreign origin and many of them have been kept more or less secret for some time. Owing to the peculiar combination of the bouquet oils and flavors, it is impossible to make accurate analyses of them. But by the use of formulas now given, these products seem to be very nearly duplicated.
It is necessary to use the best sugar and oils obtainable in the preparation of the liqueurs. As there are so many grades of essential oils on the market, it is difficult to obtain the best indirectly.
The value of the cordials is enhanced by the richness and odor and flavor of the oils, so only the best qualities should be used.
For filtering, flannel or felt is valuable. Flannel is cheaper and more easily washed. It is necessary to return filtrate several times with any of the filtering media.
As a clarifying agent talcum allowed to stand several days acts well. These rules are common to all.
[769]
WINES AND LIQUORS
The operations are all simple:
First: Heat all mixtures. Second: Keep the product in the dark. Third:
Keep in warm place.
The liqueurs are heated to ripen the bouquet flavor, it having effect similar to age. To protect the ethereal oils, air and light are excluded; hence it is recommended that the bottles be filled to the stopper. The liqueurs taste best at a temperature not exceeding 55º F. They are all improved with age, especially many of the bouquet oils.
Benedictine.
I.
Bitter almonds 40 grams
Powdered nutmeg 4.500 grams
Extract vanilla 120 grams
Powdered cloves 2 grams
Lemons, sliced 2 grams
True saffron .600 grams
Sugar 2,000 grams
Boiling milk 1,000 c.c.
Alcohol, 95 per cent 2,000 c.c.
Distilled water 2,500 c.c.
Mix. Let stand 9 days with occasional agitation. Filter sufficiently.
II.
Essence Bénédictine 75 c.c
Alcohol, 95 per cent 1,700 c.c.
Mix.
Sugar 1,750 grams
Water, distilled. 1,600 c.c.
Mix together, when clear solution of sugar is obtained. Color with aramel. Filter sufficiently.
NOTE. This liqueur should be at least 1 year old before used.
Essence Bénédictine for Bénédictine No. II.
I.
Myrrh. 1 part
Decorticated cardamom 1 part
Mace 1 part
Ginger 10 parts
Galanga root 10 parts
Extract aloe 4 parts
Alcohol 160 parts
Water 80 parts
Orange peel (cut) 10 parts
Mix, macerate 10 days and filter.
II.
Extract licorice 20 parts
Sweet spirits niter 200 parts
Acetic ether 30 parts
Spirits ammonia 1 part
Coumarin 12 parts
Vanillin 1 part
III.
Oil lemon 3 drops
Oil orange peel 3 drops
Oil wormwood 2.5 drops
Oil galangal 2 drops
Oil ginger 1 drop
Oil anise 15 drops
Oil cascarilla 15 drops
Oil bitter almond 12 drops
Oil milfoil 10 drops
Oil sassafras 7 drops
Oil angelica 6 drops
Oil hyssop 4 drops
Oil cardamom 2 drops
Oil liops 2 drops
Oil juniper 1 drop
Oil rosemary 1 drop
Mix A, B, and C.
NOTE. This essence should stand 2 years before being used for liqueurs.
Chartreuse.
I. Elixir vegetal de laGrande Chartreuse.
Fresh balm mint herbs 64 parts
Fresh hyssop herbs 64 parts
Angelica herbs and root,
fresh, together 32 parts
Cinnamon 16 parts
Saffron 4 parts
Mace 4 parts
Subject the above ingredients to maceration for a week with alcohol (96
per cent), 1,000 parts, then squeeze off and distill the liquid obtained over a certain quantity of fresh herbs of balm and hyssop. After 125 parts of sugar have been added to the resultant liqueur, filter.
The genuine Chartreuse comes in three different colors, viz., green, white, and yellow. The coloration, however, is not artificial, but is determined by the addition of varying quantities of fresh herbs in the distillation. But since it would require long and tedious trials to produce the right color in a small manufacture, the yellow shade is best imparted by a little tincture of saffron, and the green one by the addition of a few drops of indigo solution.
II.
Eau des Carmes 3 1/2 ounces
Alcohol 1 quart
Distilled water 1 quart
Sugar 1 1/2 pounds
Tincture of saffron 1 ounce
Mix. Dissolve sugar in warm water, cool, strain, add remainder of ingredients, and filter. This is known as yellow Chartreuse.
[770]
WINES AND LIQUORS
Curasao Liqueur.
A.
Oil lemon, q. s 10 drops
Oil bitter almond, q. s. 5 drops
Oil curaoa orange 15 parts
Oil sweet orange 1 part
Oil bitter orange 1 part
Cochineal 1 part
French brandy 50 parts
B.
Alcohol 4,500 parts
C.
Sugar 3,500 parts
Water (distilled) 4,000 parts
Mix A, B, and C. Filter. Color with caramel.
May Bowl or May Wine. The principal ingredient of May bowl, or that which gives it its flavor and bouquet, is fresh Waldmeisterkraut (Asperula odorata), the "woodruff" or "sweet grass," "star grass," and a dozen other aliases, of a plant growing wild all over Europe, both continental and insular, and cultivated by some gardeners in this country. It is accredited with being a diuretic, deobstruent and hepatic stimulant, of no mean order, though it has long been banished from the pharmacopoeia.
In Baden and in Bavaria in preparing Maitrank the practice was formerly to first make an essence Maitrankessenz, for the preparation of which every housewife had a formula of her own. The following was that generally used in the south of Germany:
I.
Fresh, budding woodruff, cut fine 500 parts
Alcohol, commercial (90 per cent) 1,000 parts
Digest together for 14 days, then filter and press off. Many add to this some flavoring oil. As coumarin has been found to be the principle to which the Waldmeister owes its odor, many add to the above Tonka bean, chopped fine, 1 part to the thousand. From about 12 to 15 drachms of this essence is added to make a gallon of the wine, which has about the following formula:
French brandy, say 4 drachms
Oil of unripe oranges 80 drops
Sugar 4 to 8 ounces
Essence 12 drachms
Wine to make 1 gallon
II. Take enough good woodruff (Waldmeister) of fine aroma and flavor.
Remove all parts that will not add to the excellence of the product, such as wilted, dead, or imperfect leaves, stems, etc., and wash the residue thoroughly in cold water, and with as little pressure as possible. Now choose a flask with a neck sufficiently wide to receive the stems without pressing or bruising them, and let the pieces fall into it. Pour in sufficient strong alcohol (96 per cent) to cover the herbs completely. In from 30 to 40 minutes the entire aroma is taken up by the alcohol, which takes on a beautiful green color, which, unfortunately, does not last, disappearing in a few days, but without affecting the aroma in the least. The alcohol should now be poured off, for if left to macerate longer, while it would gain in aroma, it will also take up a certain bitter principle that detracts from the delicacy of flavor and aroma. The extract is now poured on a fresh quantity of the herb, and continue proceeding in this manner until a sufficiently concentrated extract is obtained to give aroma to 100 times its weight of wine or cider.
III. Fresh woodruff, in bloom or flower, is freed from the lower part of its stem and leaves, and also of all foreign or inert matter. The herb is then lightly stuck into a wide-mouth bottle, and covered with strong alcohol. After 30 minutes pour off the liquor on fresh woodruff. In another half hour the essence is ready, though it should not be used immediately. It should be kept at cellar heat (about 60º F.) for a few days, or until the green color vanishes. Any addition to the essence of aromatics, such as orange peel, lemons, spices, etc., is to be avoided. To prepare the Maitrank, add the essence to any good white wine, tasting and testing, until the flavor suits.
The following are other formulas for the drink:
IV.
Good white wine or cider 65 parts
Alcohol, dilute 20 parts
Sugar 10 parts
Maitrankessenz 1 part
Mix.
Maraschino Liqueur.
Oil bitter almonds 15 minims
Essence vanilla 1 drachm
Jasmine extract 2 drops
Raspberry essence 10 drops
Oil neroli 10 drops
Oil lemon 15 minims
Spirits nitrous ether 2 drachms
Alcohol 6 pints
Sugar 8 pounds
Rose water 10 ounces
Water sufficient to make 2 gallons
Make a liquor in the usual manner.
To Clarify Liqueurs. For the clarification of turbid liqueurs, burnt pow-
[771]
WINES AND LIQUORS - WIRE ROPE
dered alum is frequently employed. Make a trial with 200 parts of the dim liqueur, to which 1.5 parts of burnt powdered alum is added; shake well and let stand until the liquid is clear. Then decant and filter the last portion. If the trial is successful, the whole stock may be clarified in this manner.
MEDICINAL WINES:
Beef and Iron. The following formula is recommended by the American Pharmaceutical Association:
I. Extract of beef 35 grams
Tincture of citro-chloride of iron 35 c.c.
Compound spirit of orange 1 c.c.
Hot water 60 c.c.
Alcohol 125 c.c.
Syrup 125 c.c.
Sherry wine sufficient to make 1,000 c.c.
Rub the extract of beef with the hot water, and add, while stirring, the alcohol. Allow to stand 3 days or more, then filter and distill off the alcohol. Add to the residue 750 cubic centimeters of the wine, to which the compound spirit of orange has been previously added. Finally add the tincture of citro-chloride of iron, syrup, and enough wine to make 1,000 cubic centimeters. Filter if necessary.
II. For Poultry and Stock. A good formula for wine of beef and iron is as follows:
Beef extract 256 grains
Tincture of iron citro-chloride 256 minims
Hot water 1 fluidounce
Sherry wine enough to make 1 pint
Pour the hot water in the beef extract and triturate until a smooth mixture is made. To this add, gradually and under constant stirring, 12 ounces of the wine. Add now, under same conditions, the iron, stir in well, and finally add the remainder of the wine.
Cinchona.
I. Macerate 100 parts of cinchona succirubra in coarse powder for 30 minutes in 100 parts of boiling water. Strain off the liquor and set aside. Macerate the residuum in 1,000 parts of California Malaga for 24 hours, strain off the liquid and set aside. Finally macerate the magma in 500 parts of alcohol, of 50 per cent, for 1 hour, strain off and set aside. Wash the residue with a little water to recover all the alcoholic tincture; then unite all the liquids, let stand for 24 hours, and filter. To the filtrate add 800 parts loaf sugar and dissolve by the aid of gentle heat and again filter. The product is all that could be asked of a wine of cinchona. To make a ferrated wine of this, dissolve 1 part of citro-ammoniacal pyrophosphate of iron to every 1,000 parts of wine.
II. Yvon recommends the following formula:
Red cinchona, coarse powder 5 parts
Alcohol, 60 per cent 10 parts
Diluted hydrochloric acid 1 part
Bordeaux wine 100 parts
Macerate the bark with the acid and alcohol for 6 days, shaking from time to time, add the wine, macerate for 24 hours, agitating frequently, then filter.
Removal of Musty Taste and Smell from Wine. For the removal of this
unpleasant quality, Kulisch recommends the use of a piece of charcoal of about the size of a hazel nut 5 to 10 parts per 1,000 parts of wine. After this has remained in the cask for 6 to 8 weeks, and during this time has been treated once a week with a chain or with a stirring rod, the wine can be racked off. Obstinate turbidness, as well as stalk taste and pot flavor, can also be obviated by the use of the remedy.
WINTERGREEN, TO DISTINGUISH METHYL SALICYLATE FROM OIL OF.
A quantity of the sample is mixed in a test tube with an equal volume of pure concentrated sulphuric acid. Under these conditions the artificial compound shows no rise in temperature and acquires only a slight yellowish tint, while with the natural oil there is a marked rise in temperature and the mixture assumes a rose-red color, gradually passing into darker shades.
WIRE ROPE.
See also Steel.
A valuable anti-friction and preservative compound for mine cables is as follows: Seven parts soft tallow and 3 parts plumbago, mixed thoroughly; make a long, hollow box or trough, gouge out a 4 by 6 piece of scantling about 2 feet long, sawing it down lengthwise and hollowing out the box or trough enough to hold several pounds of the compound, making also a hole lengthwise of the
[772]
WIRE ROPE - WOOD
trough for the cable to run through; then affix to rope and clamp securely, having the box or trough so fixed that it cannot play, and letting the cable pass through it while going up or down, so that it will get a thorough coating. This, it is found, will preserve a round cable very well, and can be used at least once a week. For a flat steel cable raw linseed oil can be used instead of the tallow, in about the proportion of 6 parts oil and 3 plumbago. If tar is used, linseed oil is to be added to keep the tar from adhering, both ingredients to be mixed while warm.
To preserve wire rope laid under ground, or under water, coat it with a mixture of mineral tar and fresh slaked lime in the proportion of 1 bushel of lime to 1 barrel of tar. The mixture is to be boiled, and the rope saturated with it while hot; sawdust is sometimes added to give the mixture body. Wire rope exposed to the weather is coated with raw linseed oil, or with a paint composed of equal parts of Spanish brown or lampblack with linseed oil.
WIRE HARDENING:
See Steel.
WITCH-HAZEL JELLY:
See Cosmetics.
Wood
DECORATIVE WOOD-FINISH.
Paint or stencil wood with white-lime paint. When it has dried slowly in the shade, brush it off and a handsome dark-brown tone will be imparted to the oak-wood. Some portions which may be desired darker and redder are stained again with lime, whereby these places become deeper. It is essential that the lime be applied in even thickness and dried slowly, for only then the staining will be red and uniform.
After the staining saturate the wood with a mixture of varnish, 2 parts; oil of turpentine, 1 part; turpentine, 1/2 part. When the oil ground is dry apply 2 coatings of pale amber varnish.
Colored decorations on pinewood can be produced as follows:
The most difficult part of the work is to remove the rosin accumulations without causing a spot to appear. Burn out the places carefully with a red-hot iron. Great care is necessary to prevent the iron from setting the rosin on fire, thus causing black smoke clouds.
The resulting holes are filled up with plaster to which a little light ocher is added to imitate the shade of the wood as perfectly as possible. Plaster up no more than is necessary.
Rub the wood down with very fine sandpaper, taking especial care to rub only with the grain of the wood, since all cross scratches will remain permanently visible.
After this preliminary work cover the wood with a solution of white shellac, in order not to injure the handsome golden portions of the wood and to preserve the pure light tone of the wood in general.
On this shellac ground paint and stencil with glazing colors, ground with isinglass solution. The smaller, more delicate portions, such as flowers and figures, are simply worked out in wash style with water colors, using the tone of the wood to remain as high lights, surrounding the whole with a black contour.
After this treatment the panels and decorated parts are twice varnished with dammar varnish. The friezes and pilaster strips are glazed darker and set off with stripes; to varnish them use amber varnish.
The style just mentioned does not exclude any other. Thus, for nstance, a very good effect is produced by decorating the panels only with a black covering color or with black and transparent red (burnt sienna and a little carmine) after the fashion of boule work in rich ornaments, in such a way that the natural wood forms the main part and yet quite a considerable portion of the ornament.
Intarsia imitation is likewise well adapted, since the use of ariegated covering colors is in perfect keeping with the decoration of natural wood. How it should be applied, and how much of it, depends upon one's taste, as well as the purpose and kind of the object.
It is a well-known fact that the large pores of oak always look rather smeary, according to whether the workshop is more or less dusty. If this is to be avoided, which is essential for neat work, take good wheat starch, pound it fine with a hammer and stir by means of a wooden spatula good strong polish with the wheat starch to a paste and work the paste into the pores by passing it cross-wise over the wood. After about 1/2 hour, rub down the wood thus treated in such a manner that the pores are filled. In case any open pores remain, repeat the process as before. After that, rub down, polish or deaden. If this operation is not performed, the pores will always look somewhat dirty, despite all
[773]
WOOD
care. Every cabinetmaker will readily perceive that this filling of the pores will save both time and polish in the subsequent finishing.
WOOD FILLERS.
The novice in coach painting is quite as likely to get bewildered as to be aided by much of the information given about roughstuff, the more so as the methods differ so widely. One authority tells us to use a large proportion of lead ground in oil with the coarser pigment, while another says use dry lead and but a small percentage, and still another insists that lead must be tabooed altogether. There are withal a good many moss-grown superstitions associated with the subject. Not the least of these is the remarkably absorbent nature which the surface that has been roughstuffed and "scoured" is supposed to possess. By many this power of absorption is believed to be equal to swallowing up, not only all the color applied, but at least 3 coats of varnish, and none of these would think of applying a coat of color to a roughstuffed surface without first giving it a coat of liquid filler as a sort of sacrificial oblation in recognition of this absorbing propensity. Another authority on the subject has laid down the rule that in the process of scouring, the block of pumice stone must always be moved in one direction, presumably for the reason that some trace of the stone is likely to be visible after the surface is finished.
If the block of stone is scratching, perhaps the appearance of the finished panel may be less objectionable with the furrows in parallel lines than in what engravers call "cross-hatching," but if the rubbing is properly done it is not easy to discover what difference it could make whether the stone is moved in a straight line or a circle. As to absorption, it cannot be distinguished in the finished panel between the surface that was coated with liquid filler and that to which the color was applied directly, except that cracking always occurs much sooner in the former, and this will be found to be the case with surfaces that have been coated with liquid filler and finished without roughstuff. Among the pigments that may be used for roughstuff, and there are half a dozen or more, any of which may be used with success,
there is no doubt but that known as "English filler" is best, but it is not always to be had without delay and inconveniences.
Yellow ocher, Reno umber and Keystone filler are all suitable for roughstuff, the ocher having been used many years for the purpose, but, as already remarked, the English filler is best. This is the rule for mixing given by Nobles and Hoare: Four pounds filler, 1 pound ground white lead, 1 pint gold size, 1 pint varnish and l 1/4 pints turpentine, or 3/4 pint good size and 1/2 pint boiled oil in lieu of the varnish. In regard to the use of white lead ground in oil, it makes the rubbing more laborious, increases the liability to scratching, and requires a much longer time to harden before the scouring can be done, without in any appreciable manner improving the quality of the surface when finished.
It may be remarked here that the addition of white lead, whether ground in oil or added dry to the coarser pigment, increases the labor of scouring just in proportion as it is used until sufficient may be used to render the scouring process impossible; hence, it follows that the mixing should be governed by the character of the job in hand. If the job is of a cheap class the use of very little or no lead at all is advisable, and the proportion of Japan and turpentine may also be increased, with the result that a fairly good surface may be obtained with much less labor than in the formula given.
The number of coats of filler required to effect the purpose in any given case must depend upon how well the builder has done his part of the work. If he has left the surface very uneven it follows, as a matter of course, that more coats will be required to make it level, and more of the roughstuff will remain after the leveling process than if the woodwork had been more perfectly done. While the merits of a system or method are not to be judged by its antiquity, there should be a good reason to justify the substitution of a new method for one that has given perfect satisfaction for generations and been used by the best coach painters who ever handled a brush.
A well-known writer on paints says that the effect of a varnish is usually attributed to the manner of its application and the quantity of thinners used for diluting the melted gums, with the prepared oils and the oxidizing agents used in its manufacture. While this has undoubtedly much to do with the successful application of varnish, there are other facts in this connection that should not be overlooked. For example, varnish is sometimes acted on by the breaking up, or the disintegration of the filling coats; which in turn is evidently acted on by the wood itself, according to its nature.
[774]
WOOD
With the aid of the microscope in examining the component parts of wood a cellular tissue is observed which varies in form according to the species and the parts which are inspected. This cellular tissue is made up of small cavities called pores or cells, which are filled with a widely diversified matter and are covered with a hard and usually brittle substance called lignin.
This diversified matter consists of mineral salts and various organic substances, gelatinous in their nature and held in solution by a viscous liquid and containing nitrogenous matter in different combinations, the whole being designated by the general name of albuminous substances. The older the wood the more viscous is the matter; while wood of recent growth (sapwood) contains less viscous matter holding these substances in solution. This albumen in wood acts
on substances like filler and varnish in one way or the other, good or bad. The seasoning of wood does not dispose of these substances. The water evaporates, leaving them adhering to the sides of the cells. The drier these substances are the less action they exert on the filler or whatever substance is coated on the surface. If the filler disintegrates, it affects the varnish.
All albuminous substances, be they dry or in liquid form, are subject, more or less, according to the protein they contain - which seems, or rather is, the essential principle of all albuminous matter - to the influence of caustic potash and soda. Thus, the albumen of an egg is exactly like that contained in the composition of wood. As albumen in wood becomes solid by drying, it is easily dissolved again, and will then be acted on chemically by any extraneous substance with which it comes in contact.
Some of the shellacs, substitutes for shellacs, and some of the liquid fillers are manufactured from some of the following substances: Old linseed oil, old varnish, old and hard driers, turpentine, benzine, often gasoline, rosin, whiting, cornstarch flour, hulls, paint skins, silica, and so on. The list is long. To these must be added a large volume of potash, to bring it to and hold it in solution. There must be an excess of potash which is not combined into a chemical compound, which if it did, might mitigate its influence on the albumen of the wood. But as there is potash in its pure state remaining in the solution it necessarily attacks the albumen of the wood, causing disintegration, which releases it from the wood, causing white, grayish flakes, and the formation of a powder. This is not a conclusion drawn from an inference but an established scientific fact resulting from experiments with fillers the various compositions of which were known. All alkalies act on albumen. No one would knowingly varnish over a surface such as it would be were the white of an egg applied to it and then washed with an alkali solution; but that is just what is done when varnish is put over a wood surface filled with a filler which contains an alkalI.
Most of the combinations of material used in the painting trade are mixtures; that is, each part remains the same exerting the same chemical action on another substance, or any other substance coming in contact with a paint mixture will exert the same chemical action on any part, or on any ingredient it contains, the same as if that part was by itself.
We can now account for some of the numerous peculiarities of varnish. We know that any alkali when coming in contact with albumen forms a compound, which on drying is a white, brittle substance easily disintegrated. This is why potash, sal soda, and kindred substances will remove paint. The alkali attacks the albumen in the oil, softening it, causing easy removal, whereas if it were allowed to dry, the albumen in the oil would take on a grayish color quite brittle. Potash or other alkalies in filler not only attack the albumen in the wood, but also attack the albumen in the oil by forming a compound with it. Probably this compound is very slight, only forming a compound in part, enough, nevertheless, to start a destroying influence, which is demonstrated by the following results of experiments. The reader has, perhaps, some time in his career applied a rosin varnish over a potash filler and has been surprised by the good results, a more permanent effect being obtained than in other instances where the best of varnish was used. This is accounted for by the rosin of the potash. Again, the reader may have had occasion to remove varnish with potash and found that potash would not touch it. This is because of its being a rosin varnish. Potash in filler may be rendered somewhat inert, by reason of its compounding with other parts of the filler, but owing to the quantity used in some of the commercial fillers it is not possible that all the alkali is rendered inert. Hence it will attack the albumen wherever found, as all albumen is identical in its chemical composition.
Alkalies have but little effect on the
[775]
WOOD
higher classes of gums, because of their effect on the albumen in the wood and oil. All alcohol varnishes or varnishes made by the aid of heat stand well over an alkali filler. Varnishes which contain little oil seem to stand well. This is accounted for by the fact that alcohol renders albumen insoluble. Alkalies of all kinds readily attack shellac and several other of the cheap gums, forming unstable compounds on which oil has but little effect.
Close-grained wood contains less albumen and more lignin than open-grained varieties, and consequently does not take so much filler, which accounts for the finish invariably lasting longer than the same kind used on an open-grained wood. Open-grained wood contains more sap than close grained; consequently there is more albumen to adhere to the sides of the cells. The more albumen, the more readily it is attacked by the potash, and the more readily decomposed, or rather destroyed.
Alcohol renders albumen insoluble immediately on application. It prevents it from compounding with any other substance, or any other substance compounding with it. Hence, we must conclude that an application of alcohol to wood before the filler is applied is valuable, which is proven to be a fact by experiment. Wash one half of a board with alcohol, then apply the potash filler over all. Again, wash the portion of the board on which is the filler and apply a heavy-bodied oil varnish. Expose to sunlight and air the same as a finished door or the like, and wait for the result. At the end of a few months a vast difference will be found in the two parts of the surface. The one on which there is no alcohol will show the ravages of time and the elements much sooner than the one on which it is.
Wood finishers demand a difference in the composition of fillers, paste and liquid, for open- and close-grained wood, respectively; but unfortunately they do not demand a difference between either kind in themselves, according to the kind of wood. Paste fillers are used indiscriminately for open-grained wood and liquid for close-grained wood.
To find the fillers best adapted for a certain wood, and to classify them in this respect will require a large amount of chemical work and practical experiments; but that it should be done is evidenced by the fact that both success and failure result from the use of the same filler on different varieties of wood. After once being classified (owing to the large number now on the market), they will not number nearly so many in the aggregate as might be supposed; as it will be found in many instances that two entirely different varieties of wood resemble each other more closely in their vascular formation and cell characteristics than do two other specimens of the same variety.
It is a recognized fact that paste fillers whose base is starch or the like work better and give better results in certain instances, while those whose base is mineral matter seem to do better in other cases.
It is noticed that rosewood as a finishing veneer is obsolete. This is not because of its scarcity, but because it is so hard to finish without having been seasoned for a long time. In these days, manufacturers cannot wait. It takes longer for the sap of rosewood to become inactive, or in trade parlance to "die," than any other wood. This is because it takes so long for the albumen in the sap to coagulate. Rosewood has always been a source of trouble to piano makers, on account of the action of the sap on the varnish. However, if this wood, previously to filling, was washed with a weak solution of phosphoric acid, and then with wood spirit, it might be more easily finished. The phosphoric acid would coagulate the albumen on the surface of the wood immediately, while alcohol would reduce it to an insoluble state. The idea here is to destroy the activity of the sap, on the same principle as sappy places and knot sap are destroyed by alcohol-shellac before being painted.
Oak is another wood which gives the painter trouble to finish. This may be accounted for as follows: Oak contains a sour acid principle called tannic acid. It is a very active property. Wood during the growing season contains more albumen; thus in the circulation of the sap a large quantity of soft matter is deposited on the lignin which lines the cells, which lignin, if it contains any acid matter, acts on the material of the filler. Tannic acid has a deleterious effect on some of the material of which a number of fillers are made. Starch and many gums are susceptible to its influence, making some of them quite soft. Oak, like most other timber cut at the season when the least sap is in circulation, is the more easily finished.
The vascular formation may, and no doubt has, something to do with wood finishing. Different species of wood differ materially in their vascular and cellular formation. Wood finishers recognize a difference in treatment of French burl walnut and the common American